
BITSAT 2024 Notification, Application Form, Exam Dates, Eligibility, Pattern
BITS Pilani invites applications to register for BITSAT-2024, the only online admission test for admissions to Integrated first degree (B.E., B.Pharm, and M.Sc.) programs in its campuses at Pilani, Goa, and Hyderabad. BITSAT 2024 serves as the gateway for admission to BITS for various engineering and science programs at undergraduate and postgraduate level. The BITSAT 2024 or the Birla Institute of Technology and Science Admission Test, is a prestigious computer-based entrance examination in India, conducted by BITS Pilani. BITSAT 2024 assesses candidates on their proficiency in subjects like mathematics, physics, and chemistry, along with an additional section on English proficiency and logical reasoning. BITSAT 2024 is conducted during May-July every year in 2 sessions. BITSAT 2024 is will take place on May 19-24, 2024 and June 22-26, 2024 for Session-I and II, respectively.
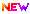 BITSAT 2024 Application Form has been released from January 15, 2024. Check here for application form details.
BITSAT 2024 Application Form has been released from January 15, 2024. Check here for application form details.
BITSAT 2024 Exam Pattern & Syllabus has been released through online mode. Check BITSAT 2024 Exam Pattern details here.
BITSAT 2024 Exam Date has been announced. The exam for Session-1 will be held from May 19-24, 2024 and for Session-2 from June 22-26, 2024.
What is BITSAT?
The BITSAT 2024 is an entrance examination conducted by BITS Pilani, one of the most prestigious technical universities in India. BITSAT 2024 is specifically designed for admission to various undergraduate engineering and science programs offered by BITS at various campuses. It is a computer-based test that assesses candidates in subjects like mathematics, physics, and chemistry, along with an additional section on English proficiency and logical reasoning.
BITSAT 2024 is conducted in 2 sessions. The candidates can attend either of the sessions or both the sessions with the application fee for each of them varying accordingly. BITSAT is a computer-based test, which means that candidates take the exam on a computer at designated test centers. The BITSAT 2024 is three hours long with the medium of instruction as English.
Courses Offered by various Campuses
at BITS Pilani – Pilani Campus:
- B.E.: Chemical, Civil, Computer Science, Electrical and Electronics, Electronics & Instrumentation, Electronics & Communication, Mechanical, Manufacturing.
- B. Pharm.
- M.Sc*.: Biological Sciences, Chemistry, Economics, Mathematics, Physics
- M.Sc.: General Studies.
at BITS Pilani – K. K. Birla Goa Campus:
- B.E.: Chemical, Computer Science, Electronics & Communication, Electrical and Electronics, Electronics & Instrumentation, Mechanical, Mathematics and Computing.
- M.Sc*.: Biological Sciences, Chemistry, Economics, Mathematics, and Physics.
at BITS Pilani – Hyderabad Campus:
- B.E.: Chemical, Civil, Computer Science, Electronics & Communication, Electrical and Electronics, Electronics & Instrumentation, Mechanical, Mathematics and Computing.
- B. Pharm.
- M.Sc*.: Biological Sciences, Chemistry, Economics, Mathematics, and Physics.
- *The candidates who join in M.Sc. programme are eligible to apply for dual degree in Engineering at BITS Pilani after completion of first year. This assignment is made by competition on their performance at BITS at the end of the first year. (For more details refer to Admissions Modality available on the BITS admissions website.)
BITSAT 2024 Exam Date
- Start of application form : January 15, 2024
- Deadline to apply online for BITSAT-2024 for Session-1 or Both the sessions : April 11, 2024
- Revision/editing (online) in the application form by candidates : April 15-19, 2024
- Test center allotment and announcement to candidates : May 1, 2024
- Candidates to reserve Test date and slot. $ : May 6-10, 2024
- Candidates to download Hall tickets with instructions (for Session-1) : May 15, 2024 to till the exam date
- BITSAT-2024 Online Test Session-1* : May 19-24, 2024
- The application window to apply for BITSAT-2024 Session-2 only : May 22 to June 10, 2024
- Revision/editing (online) in the application form by candidates : June 11-12, 2024
- Test center allotment and announcement to candidates : June 13, 2024
- Candidates to reserve Test date and slot. : June 15-17, 2024
- Candidates to download the Hall tickets with instructions : June 19, 2024 to till the exam date
- BITSAT-2024 Online Test Session-2* : June 22-26, 2024
- Apply for admission with 12th Marks and Preferences of Programmes : June 1-28, 2024
- Editing of Marks/Preferences in Application form : June 29-30, 2024
- Dates and No. of days may vary from center to center (These dates are tentative)
BITSAT 2024 Eligibility Criteria
The following is the BITSAT 2024 Eligibility Criteria:-
| Eligibility Criteria |
Details |
| 1. Educational Qualifications |
- Candidates must have passed the 12th examination from a recognized Central or State board or its equivalent.
- For most programs, the 12th examination should include Physics, Chemistry, and Mathematics, along with proficiency in English.
- For the B. Pharm. program, candidates should have studied Physics, Chemistry, and Biology, along with English proficiency. However, candidates with Physics, Chemistry, and Mathematics (PCM) can also apply for the Pharmacy program.
|
| 2. Minimum Marks Requirement |
- Candidates with PCM subjects must secure a minimum of 75% aggregate marks in the 12th examination.
- In the 12th examination, candidates must have at least 60% marks in each of the Physics, Chemistry, and Mathematics (or Biology) subjects.
|
| 3. Applicable Year |
- Only students who are appearing for the 12th examination in 2024 or have passed it in 2023 are eligible for BITSAT-2024.
- Candidates with multiple attempts in the 12th class are considered based on their latest performance, provided it covers all required subjects.
- Students who passed the 12th examination in 2022 or earlier are not eligible.
- Current BITS students cannot appear in BITSAT-2024.
|
| 4. Admission Criteria |
- Admission is solely based on merit, determined by the BITSAT-2024 score.
- Merit candidates must also meet the minimum marks requirement in their 12th examination, as specified.
|
| 5. Direct Admission for Board Toppers |
- Students who secure the first rank in their respective board examinations for 2024 will be granted direct admission to their chosen program, irrespective of their BITSAT-2024 score, provided they meet the eligibility criteria.
|
| 6. Age limit |
- There is no age criteria for participating in the BITSAT 2024 admission process.
|
BITSAT 2024 Application Form
The BITSAT 2024 Application Form has been released. Here is the process to fill the BITSAT 2024 Application Form:-
Step 1- BITSAT Registration
- The initial step involves creating a new account. Candidates are required to provide a valid email address and mobile number for this purpose.
Step 2- Form Filling
- Once the registration is completed, candidates must proceed to fill out the BITSAT application form for 2024.
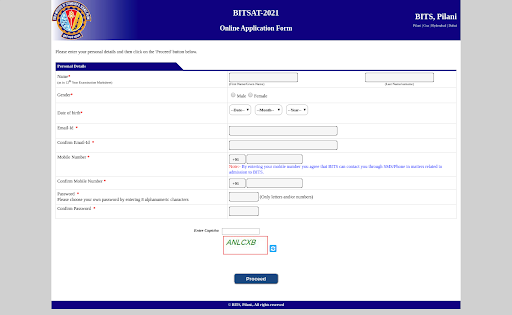
- During this stage, applicants need to furnish personal, contact, and academic details.
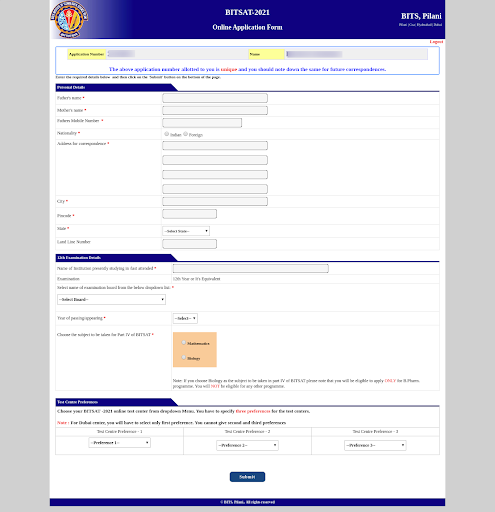
- Additionally, they should specify their preferred examination center. It’s important to select at least three preferred exam centers.
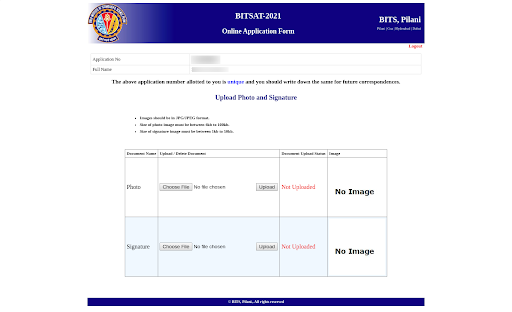
Step 3- Uploading Documents
- While filling out the application form, candidates will need to upload scanned copies of their photograph and signature in the specified format.
Step 4- Fee Payment
- The candidate at last needs to complete the BITSAT 2024 Application Process by paying the fee via PayUMoney, Paytm or ICICI Bank.
BITSAT 2024 Application Form- Documents Required
To streamline the BITSAT 2024 application process, it is recommended that candidates prepare all the necessary documents in advance. The following are the documents essential when filling out the BITSAT 2024 Application Form:-
- Academic records, including statements of marks for both Class 10 and 12. If a candidate is presently in Class 12, only the Class 10 marks statement is required.
- Scanned copies of passport-sized photographs and signatures, adhering to the specified image requirements.
- Credit or debit card details for the payment of BITSAT 2024 registration fees.
BITSAT 2024 Application Form- Image Specifications
| Type of Image |
Permissible Size |
Permissible Format |
| Photograph |
4 Kb to 100 Kb |
JPEG/JPG |
| Signature |
1 Kb to 40 Kb |
JPEG/JPG |
BITSAT 2024 Application Fee
The following is the BITSAT 2024 Application Fee:-
| Category |
Session 1 |
Session 2 (If appeared for session 1; Additional Fee) |
Both Sessions |
| Male Candidates |
INR 3,400/- |
INR 2000 |
INR 5,400/- |
| Female Candidates |
INR 2,900/- |
INR 1500 |
INR 4,400/- |
| For Dubai Centre (Male) |
INR 7,000/- |
INR 2000 |
INR 9,000 |
| For Dubai Centre (Female) |
INR 9,000/- |
INR 2000 |
INR 9,000 |
It is to be noted that, If a candidate initially selects to take the BITSAT during Session 1 and later decides to apply separately for a second attempt in Session 2, they will be required to pay an additional fee of Rs. 2000 for male candidates and Rs. 1500 for female candidates.
The application fee cannot be refunded or transferred to another candidate.In the case of a candidate who participates in BITSAT-2024 on two separate occasions, the higher of the two scores will be taken into consideration when seeking admission to any of the First Degree programs at BITS Pilani.
BITSAT 2024 Exam Pattern
Each BITSAT-2024 session will last for 3 hours, without any breaks.The exam is divided into four parts as given in the following table:-
| Part |
Subject |
No of Questions |
| I |
Physics |
30 |
| II |
Chemistry |
30 |
| III |
(A) English Proficiency
(B) Logical Reasoning |
(A) 10
(B) 20 |
| IV |
Biology/ Mathematics (For B. Pharm) |
40 |
| Total |
130 |
- Question Types: All questions are of the objective type (multiple choice questions). Each question has four answer options, with only one being the correct choice. Correct answers earn 3 marks, while incorrect answers result in a penalty of 1 mark (-1 mark). No marks are awarded for questions left unanswered.
- Question Count: There will be a total of 130 questions. The distribution of questions in each part is as follows. There are no time limits for individual parts, and candidates can navigate freely among the 130 questions.
- Extra Questions: If a candidate answers all 130 questions without skipping any, they will have the option to attempt 12 additional questions (maximum) if time remains. These extra questions will be drawn from Physics, Chemistry, Mathematics/Biology, and Logical reasoning, with three questions from each category. Once a candidate opts for extra questions, they cannot revise their earlier 130 answers.
- Question Selection: The questions are designed so that a proficient student can answer all 130 questions within the 180-minute time frame. Extra questions (up to 12) offer a chance for highly meritorious candidates to score higher, but it’s important to note that there is negative marking for incorrect answers. Blind guessing is not advised, as it may lead to a reduction in the total score.
- Randomization: The questions are selected randomly from a vast question bank. Different candidates receive different sets of questions. An expert committee ensures that the question sets have comparable difficulty levels, content, and question types. The committee’s decisions are final and binding.
- Language: All questions and instructions are in English.
BITSAT 2024 Syllabus
BITSAT 2024 will follow the curriculum outlined in the NCERT syllabus for the 11th and 12th grades. A comprehensive syllabus can be found on https://www.bitsadmission.com/bitsat/download/BITSAT-2023_brochure.pdf?31012023 , and candidates are encouraged to consult NCERT textbooks for the specific content.
For the ease of the candidate we are providing you with the detailed BITSAT 2024 Syllabus.
BITSAT 2024 Physics Syllabus
The following is the BITSAT 2024 Physics Syllabus:-
| Topics |
| Units & Measurement |
- Units (Different systems of units, SI units, fundamental and derived units)
- Dimensional Analysis
- Precision and significant figures
- Fundamental measurements in Physics (Vernier calipers, screw gauge, Physical balance etc)
|
| Kinematics |
- Properties of vectors
- Position, velocity and acceleration vectors
- Motion with constant acceleration
- Projectile motion
- Uniform circular motion
- Relative motion
|
| Newton’s Laws of Motion |
- Newton’s laws (free body diagram, resolution of forces)
- Motion on an inclined plane
- Motion of blocks with pulley systems
- Circular motion – centripetal force
- Inertial and non-inertial frames
|
| Impulse and Momentum |
- Definition of impulse and momentum
- Conservation of momentum
- Collisions
- Momentum of a system of particles
- Center of mass
|
| Work and Energy |
- Work done by a force
- Kinetic energy and work-energy theorem
- Power
- Conservative forces and potential energy
- Conservation of mechanical energy
|
| Rotational Motion |
- Description of rotation (angular displacement, angular velocity and angular acceleration)
- Rotational motion with constant angular acceleration
- Moment of inertia, Parallel and perpendicular axes theorems, rotational kinetic energy
- Torque and angular momentum
- Conservation of angular momentum
- Rolling motion
|
| Gravitation |
- Newton’s law of gravitation
- Gravitational potential energy, Escape velocity
- Motion of planets – Kepler’s laws, satellite motion
|
| Mechanics of Solids and Fluids |
- Elasticity
- Pressure, density and Archimedes’ principle
- Viscosity and Surface Tension
- Bernoulli’s theorem
|
| Oscillations |
- Kinematics of simple harmonic motion
- Spring mass system, simple and compound pendulum
- Forced & damped oscillations, resonance
|
| Waves |
- Progressive sinusoidal waves
- Standing waves in strings and pipes
- Superposition of waves, beats
- Doppler Effect
|
| Heat and Thermodynamics |
- Kinetic theory of gasses
- Thermal equilibrium and temperature
- Specific heat, Heat Transfer – Conduction, convection and radiation, thermal conductivity, Newton’s law of cooling Work, heat and first law of thermodynamics
- Second law of thermodynamics, Carnot engine – Efficiency and Coefficient of performance
|
| Electrostatics |
- Coulomb’s law
- Electric field (discrete and continuous charge distributions)
- Electrostatic potential and Electrostatic potential energy
- Gauss’ law and its applications
- Electric dipole
- Capacitance and dielectrics (parallel plate capacitor, capacitors in series and parallel)
|
| Current Electricity |
- Ohm’s law, Joule heating
- DC circuits – Resistors and cells in series and parallel, Kirchoff’s laws, potentiometer, and Wheatstone bridge
- Electrical Resistance (Resistivity, origin and temperature dependence of resistivity)
|
| Magnetic Effect of Current |
- Biot-Savart’s law and its applications
- Ampere’s law and its applications
- Lorentz force, force on current-carrying conductors in a magnetic field
- Magnetic moment of a current loop, torque on a current loop, Galvanometer and its conversion to voltmeter and ammeter
|
| Electromagnetic Induction |
- Faraday’s law, Lenz’s law, eddy currents
- Self and mutual inductance
- Transformers and generators
- Alternating current (peak and rms value)
- AC circuits, LCR circuits
|
| Optics |
- Laws of reflection and refraction
- Lenses and mirrors
- Optical instruments – telescope and microscope
- Interference – Huygen’s principle, Young’s double slit experiment
- Interference in thin films
- Diffraction due to a single slit
- Electromagnetic waves and their characteristics (only qualitative ideas), Electromagnetic spectrum
- Polarization – states of polarization, Malus’ law, Brewster’s law
|
| Modern Physics |
- Dual nature of light and matter – Photoelectric effect, De Broglie wavelength
- Atomic models – Rutherford’s experiment, Bohr’s atomic model
- Hydrogen atom spectrum
- Radioactivity
- Nuclear reactions : Fission and fusion, binding energy
|
| Electronic Devices |
- Energy bands in solids (qualitative ideas only), conductors, insulators and semiconductors;
- Semiconductor diode – I-V characteristics in forward and reverse bias, diode as a rectifier; I-V characteristics of LED, photodiode, solar cell, and Zener diode; Zener diode as a voltage regulator.
- Junction transistor, transistor action, characteristics of a transistor; transistor as an amplifier (common emitter configuration) and oscillator
- Logic gates (OR, AND, NOT, NAND and NOR). Transistor as a switch
|
BITSAT 2024 Chemistry Syllabus
The following is the BITSAT 2024 Chemistry Syllabus:-
| Topics |
| States of Matter |
- Measurement: Physical quantities and SI units, Dimensional analysis, Precision, Significant figures.
- Chemical reactions: Laws of chemical combination, Dalton’s atomic theory; Mole concept; Atomic, molecular and molar masses; Percentage composition empirical & molecular formula; Balanced chemical equations & stoichiometry
- Three states of matter, intermolecular interactions, types of bonding, melting and boiling points Gaseous state: Gas Laws, ideal behavior, ideal gas equation, empirical derivation of gas equation, Avogadro number, Deviation from ideal behavior – Critical temperature, Liquefaction of gasses, van der Waals equation.
- Liquid state: Vapour pressure, surface tension, viscosity.
- Solid state: Classification; Space lattices & crystal systems; Unit cell in two dimensional and three dimensional lattices, calculation of density of unit cell – Cubic & hexagonal systems; Close packing; Crystal structures: Simple AB and AB2 type ionic crystals, covalent crystals – diamond & graphite, metals. Voids, number of atoms per unit cell in a cubic unit cell, Imperfections- Point defects, non-stoichiometric crystals; Electrical, magnetic and dielectric properties; Amorphous solids – qualitative description. Band theory of metals, conductors, semiconductors and insulators, and n- and p- type semiconductors.
|
| Atomic Structure |
- Introduction: Subatomic particles; Atomic number, isotopes and isobars, Thompson’s model and its limitations, Rutherford’s picture of atoms and its limitations; Hydrogen atom spectrum and Bohr model and its limitations.
- Quantum mechanics: Wave-particle duality – de Broglie relation, Uncertainty principle; Hydrogen atom: Quantum numbers and wavefunctions, atomic orbitals and their shapes (s, p, and d), Spin quantum number.
- Many electron atoms: Pauli exclusion principle; Aufbau principle and the electronic configuration of atoms, Hund’s rule.
|
| Periodicity: Brief history of the development of periodic tables Periodic law and the modern periodic table; Types of elements: s, p, d, and f blocks; Periodic trends: ionization energy, atomic, and ionic radii, inter gas radii, electron affinity, electronegativity and valency. Nomenclature of elements with atomic number greater than 100.Chemical Bonding & Molecular Structure |
- Valence electrons, Ionic Bond: Lattice Energy and Born-Haber cycle; Covalent character of ionic bonds and polar character of covalent bond, bond parameters
- Molecular Structure: Lewis picture & resonance structures, VSEPR model & molecular shapes
- Covalent Bond: Valence Bond Theory- Orbital overlap, Directionality of bonds & hybridization (s, p & d orbitals only), Resonance; Molecular orbital theory- Methodology, Orbital energy level diagram, Bond order, Magnetic properties for homonuclear diatomic species (qualitative idea only).
- Dipole moments; Hydrogen Bond.
|
| Thermodynamics |
- Basic Concepts: Systems and surroundings; State functions; Intensive & Extensive Properties; Zeroth Law and Temperature
- First Law of Thermodynamics: Work, internal energy, heat, enthalpy, heat capacities and specific heats, measurements of ΔU and ΔH, Enthalpies of formation, phase transformation, ionization, electron gain; Thermochemistry; Hess’s Law, Enthalpy of bond dissociation, combustion, atomization, sublimation, solution and dilution
- Second Law: Spontaneous and reversible processes; entropy; Gibbs free energy related to spontaneity and non-spontaneity, non-mechanical work; Standard free energies of formation, free energy change and chemical equilibrium
- Third Law: Introduction
|
| Physical and Chemical Equilibria |
- Concentration Units: Mole Fraction, Molarity, and Molality
- Solutions: Solubility of solids and gasses in liquids, Vapour Pressure, Raoult’s law, Relative lowering of vapor pressure, depression in freezing point; elevation in boiling point; osmotic pressure, determination of molecular mass; solid solutions, abnormal molecular mass, van’t Hoff factor. Equilibrium: Dynamic nature of equilibrium, law of mass action
- Physical Equilibrium: Equilibria involving physical changes (solid-liquid, liquid-gas, solid-gas), Surface chemistry, Adsorption, Physical and Chemical adsorption, Langmuir Isotherm, Colloids and emulsion, classification, preparation, uses.
- Chemical Equilibria: Equilibrium constants (KP, KC), Factors affecting equilibrium, Le- Chatelier’s principle.
- Ionic Equilibria: Strong and Weak electrolytes, Acids and Bases (Arrhenius, Lewis, Lowry and Bronsted) and their dissociation; degree of ionization, Ionization of Water; ionization of polybasic acids, pH; Buffer solutions; Henderson equation, Acid-base titrations; Hydrolysis; Solubility Product of Sparingly Soluble Salts; Common Ion Effect.
- Factors Affecting Equilibria: Concentration, Temperature, Pressure, Catalysts, Significance of ΔG and ΔG0 in Chemical Equilibria.
|
| Electrochemistry |
- Redox Reactions: Oxidation-reduction reactions (electron transfer concept); Oxidation number; Balancing of redox reactions; Electrochemical cells and cell reactions; Standard electrode potentials; EMF of Galvanic cells; Nernst equation; Factors affecting the electrode potential; Gibbs energy change and cell potential; Secondary cells; dry cells, Fuel cells; Corrosion and its prevention.
- Electrolytic Conduction: Electrolytic Conductance; Specific and molar conductivities; variations of conductivity with concentration , Kolhrausch’s Law and its application, Electrolysis, Faraday’s laws of electrolysis; Electrode potential and electrolysis, Commercial production of the chemicals, NaOH, Na, Al.
|
| Chemical Kinetics |
- Aspects of Kinetics: Rate and Rate expression of a reaction; Rate constant; Order and molecularity of the reaction; Integrated rate expressions and half-life for zero and first order reactions.
- Factor Affecting the Rate of the Reactions: Concentration of the reactants, catalyst; size of particles, Temperature dependence of rate constant concept of collision theory (elementary idea, no mathematical treatment); Activation energy.
- Surface Chemistry: Adsorption – physisorption and chemisorption; factors affecting adsorption of gasses on solids; catalysis: homogeneous and heterogeneous, activity and selectivity: enzyme catalysis, colloidal state: distinction between true solutions, colloids and suspensions; lyophilic, lyophobic multi molecular and macromolecular colloids; properties of colloids; Tyndall effect, Brownian movement, electrophoresis, coagulations; emulsions–types of emulsions.
|
| Hydrogen and s-block elements |
- Hydrogen: Element: unique position in periodic table, occurrence, isotopes; Dihydrogen: preparation, properties, reactions, and uses; Molecular, saline, ionic, covalent, interstitial hydrides; Water: Properties; Structure and aggregation of water molecules; Heavy water; Hydrogen peroxide: preparation, reaction, structure & use, Hydrogen as a fuel.
- S-block elements: Abundance and occurrence; Anomalous properties of the first elements in each group; diagonal relationships; trends in the variation of properties (ionization energy, atomic & ionic radii).
- Alkali metals: Lithium, sodium and potassium: occurrence, extraction, reactivity, and electrode potentials; Biological importance; Reactions with oxygen, hydrogen, halogens water; Basic nature of oxides and hydroxides; Halides; Properties and uses of compounds such as NaCl, Na2CO3, NaHCO3, NaOH, KCl, and KOH.
- Alkaline earth metals: Magnesium and calcium: Occurrence, extraction, reactivity and electrode potentials; Reactions with O2, H2O, H2 and halogens; Solubility and thermal stability of oxo salts; Biological importance of Ca and Mg; Preparation, properties and uses of important compounds such as CaO, Ca(OH)2, plaster of Paris, MgSO4, MgCl2, CaCO3, and CaSO4.
|
| p, d, and f-block elements |
- General: Abundance, distribution, physical and chemical properties, isolation and uses of elements; Trends in chemical reactivity of elements of a group; electronic configuration, oxidation states; anomalous properties of first element of each group.
- Group 13 elements: Boron; Properties and uses of borax, boric acid, boron hydrides & halides. Reaction of aluminum with acids and alkalis;
- Group 14 elements: Carbon: carbon catenation, physical & chemical properties, uses, allotropes (graphite, diamond, fullerenes), oxides, halides and sulfides, carbides; Silicon: Silica, silicates, silicone, silicon tetrachloride, Zeolites, and their uses
- Group 15 elements: Dinitrogen; Preparation, reactivity and uses of nitrogen; Industrial and biological nitrogen fixation; Compound of nitrogen; Ammonia: Haber’s process, properties and reactions; Oxides of nitrogen and their structures; Properties and Ostwald’s process of nitric acid production; Fertilizers – NPK type; Production of phosphorus; Allotropes of phosphorus; Preparation, structure and properties of hydrides, oxides, oxoacids (elementary idea only) and halides of phosphorus, phosphine.
- Group 16 elements: Isolation and chemical reactivity of dioxygen; Acidic, basic and amphoteric oxides; Preparation, structure and properties of ozone; Allotropes of sulfur; Preparation/production properties and uses of sulfur dioxide and sulphuric acid; Structure and properties of oxides, oxoacids (structures only).
- Group 17 and group 18 elements: Structure and properties of hydrides, oxides, oxoacids of halogens (structures only); preparation, properties & uses of chlorine & HCl; Interhalogen compounds; Bleaching Powder; Uses of Group 18 elements, Preparation, structure and reactions of xenon fluorides, oxides, and oxoacids.
- d-Block elements: General trends in the chemistry of first row transition elements; Metallic character; Oxidation state; ionization enthalpy; Ionic radii; Color; Catalytic properties; Magnetic properties; Interstitial compounds; Occurrence and extraction of iron, copper, silver, zinc, and mercury; Alloy formation; Steel and some important alloys; preparation and properties of K2Cr2O7, KMnO4.
- f-Block elements: Lanthanoids and actinoids; Oxidation states and chemical reactivity of lanthanide compounds; Lanthanide contraction and its consequences, Comparison of actinoids and lanthanoids.
- Coordination Compounds: Coordination number; Ligands; Werner’s coordination theory; IUPAC nomenclature; Application and importance of coordination compounds (in qualitative analysis, extraction of metals and biological systems e.g. chlorophyll, vitamin B12, and hemoglobin); Bonding: Valence-bond approach, Crystal field theory (qualitative); Isomers including stereoisomers.
|
| Principles of Organic Chemistry and Hydrocarbons |
- Classification: General Introduction, classification based on functional groups, trivial and IUPAC nomenclature. Methods of purification: qualitative and quantitative,
- Electronic displacement in a covalent bond: Inductive, resonance effects, and hyperconjugation; free radicals; carbocations, carbanions, nucleophiles and electrophiles; types of organic reactions, free radical halogenations.
- 10.3 Alkanes: Structural isomerism, general properties and chemical reactions, free radical halogenation, combustion and pyrolysis.
- Alkenes and alkynes: General methods of preparation and reactions, physical properties, electrophilic and free radical additions, acidic character of alkynes and (1,2 and 1,4) addition to dienes.
- Aromatic hydrocarbons: Sources; properties; isomerism; resonance delocalization; aromaticity; polynuclear hydrocarbons; IUPAC nomenclature; mechanism of electrophilic substitution reaction, directive influence and effect of substituents on reactivity; carcinogenicity and toxicity.
- Haloalkanes and haloarenes: Physical properties, nomenclature, optical rotation, chemical reactions and mechanism of substitution reaction. Uses and environmental effects; di, tri, tetrachloroethane, iodoform, freon and DDT.
|
| Stereochemistry |
- Conformations: Ethane conformations; Newman and Sawhorse projections.
- Geometrical isomerism in alkenes
|
| Organic Compounds with Functional Groups Containing Oxygen and Nitrogen |
- General: Nomenclature, electronic structure, important methods of preparation, identification, important reactions, physical and chemical properties, uses of alcohols, phenols, ethers, aldehydes, ketones, carboxylic acids, nitro compounds, amines, diazonium salts, cyanides and isocyanides.
- Specific: Reactivity of -hydrogen in carbonyl compounds, effect of substituents on alpha- carbon on acid strength, comparative reactivity of acid derivatives, mechanism of nucleophilic addition and dehydration, basic character of amines, methods of preparation, and their separation, importance of diazonium salts in synthetic organic chemistry.
|
| Biological , Industrial and Environmental chemistry |
- Carbohydrates: Classification; Monosaccharides; Structures of pentoses and hexoses; Simple chemical reactions of glucose, Disaccharides: reducing and non-reducing sugars – sucrose, maltose and lactose; Polysaccharides: elementary idea of structures of starch, cellulose and glycogen.
- Proteins: Amino acids; Peptide bond; Polypeptides; Primary structure of proteins; Simple idea of secondary , tertiary and quaternary structures of proteins; Denaturation of proteins and enzymes.
- Nucleic Acids: Types of nucleic acids; Primary building blocks of nucleic acids (chemical composition of DNA & RNA); Primary structure of DNA and its double helix; Replication; Transcription and protein synthesis; Genetic code.
- Vitamins: Classification, structure, functions in biosystems; Hormones
- Polymers: Classification of polymers; General methods of polymerization; Molecular mass of polymers; Biopolymers and biodegradable polymers; methods of polymerization (free radical, cationic and anionic addition polymerizations); Copolymerization: Natural rubber; Vulcanization of rubber; Synthetic rubbers. Condensation polymers.
- Pollution: Environmental pollutants; soil, water and air pollution; Chemical reactions in atmosphere; Smog; Major atmospheric pollutants; Acid rain; Ozone and its reactions; Depletion of ozone layer and its effects; Industrial air pollution; Greenhouse effect and global warming; Green Chemistry, study for control of environmental pollution.
- Chemicals in medicine, health-care and food: Analgesics, Tranquilizers, antiseptics, disinfectants, antimicrobials, anti-fertility drugs, antihistamines, antibiotics, antacids; Preservatives, artificial sweetening agents, antioxidants, soaps and detergents.
|
| 14. Theoretical Principles of Experimental Chemistry |
- Volumetric Analysis: Principles; Standard solutions of sodium carbonate and oxalic acid; Acid- base titrations; Redox reactions involving KI, H2SO4, Na2SO3, Na2S2O3 and H2S; Potassium permanganate in acidic, basic and neutral media; Titrations of oxalic acid, ferrous ammonium sulfate with KMnO4, K2Cr2O7/Na2S2O3, Cu(II)/Na2S2O3.
- Qualitative analysis of Inorganic Salts: Principles in the determination of the cations Pb2+, Cu2+, As3+, Mn2+, Al3+, Zn2+, Co2+, Ca2+, Sr2+, Ba2+, Mg2+, NH4+, Fe3+, Ni2+ and the anions CO32-, S2-, SO42-, SO32-, NO2-, NO3-, Cl-, Br-, I-, PO43-, CH3COO-, C2O42-.
- Physical Chemistry Experiments: preparation and crystallization of alum, copper sulfate. Benzoic acid ferrous sulfate, double salt of alum and ferrous sulfate, potassium ferric sulfate; Temperature vs. solubility; Study of pH charges by common ion effect in case of weak acids and weak bases; pH measurements of some solutions obtained from fruit juices, solutions of known and varied concentrations of acids, bases and salts using pH paper or universal indicator; Lyophilic and lyophobic sols; Dialysis; Role of emulsifying agents in emulsification. Equilibrium studies involving ferric and thiocyanate ions (ii) [Co(H2O)6]2+ and chloride ions; Enthalpy determination for strong acid vs. strong base neutralization reaction (ii) hydrogen bonding interaction between acetone and chloroform; Rates of the reaction between (i) sodium thiosulphate and hydrochloric acid, (ii) potassium iodate and sodium sulphite (iii) iodide vs. hydrogen peroxide, concentration and temperature effects in these reactions.
- Purification Methods: Filtration, crystallization, sublimation, distillation, differential extraction, and chromatography. Principles of melting point and boiling point determination; principles of paper chromatographic separation – Rf values.
- Qualitative Analysis of Organic Compounds: Detection of nitrogen, sulfur, phosphorous and halogens; Detection of carbohydrates, fats and proteins in foodstuff; Detection of alcoholic, phenolic, aldehydic, ketonic, carboxylic, amino groups and unsaturation.
- Quantitative Analysis of Organic Compounds: Basic principles for the quantitative estimation of carbon, hydrogen, nitrogen, halogen, sulfur and phosphorus; Molecular mass determination by silver salt and chloroplatinate salt methods; Calculations of empirical and molecular formulae.
- Principles of Organic Chemistry Experiments: Preparation of iodoform, acetanilide, p-nitro acetanilide, di-benzyl acetone, aniline yellow, beta-naphthol; Preparation of acetylene and study of its acidic character.
- Basic Laboratory Technique: Cutting glass tube and glass rod, bending a glass tube, drawing out a glass jet, boring of cork.
|
BITSAT 2024 Mathematics Syllabus
The following is the BITSAT 2024 Mathematics Syllabus:-
| Topics |
| Algebra |
- Complex numbers, addition, multiplication, conjugation, polar representation, properties of modulus and principal argument, triangle inequality, roots of complex numbers, geometric interpretations; Fundamental theorem of algebra.
- Theory of Quadratic equations, quadratic equations in real and complex number systems and their solutions.
- Arithmetic and geometric progressions, arithmetic, geometric and arithmetico• geometric series, sums of finite arithmetic and geometric progressions, infinite geometric series, sums of squares and cubes of the first n natural numbers.
- Logarithms and their properties.
- Exponential series.
- Permutations and combinations, Permutations as an arrangement and combination as selection, simple applications.
- Binomial theorem for a positive integral index, properties of binomial coefficients, Pascal’s triangle
- Matrices and determinants of order two or three, properties and evaluation of determinants, addition and multiplication of matrices, adjoint and inverse of matrices, Solutions of simultaneous linear equations in two or three variables, elementary row and column operations of matrices, Types of matrices, applications of determinants in finding the area of triangles.
- Sets, Relations and Functions, algebra of sets applications, equivalence relations, mappings, one•one, into and onto mappings, composition of mappings, binary operation, inverse of function, functions of real variables like polynomial, modulus, signum and greatest integer.
- Mathematical reasoning and methods of proofs, Mathematically acceptable statements. Connecting words/phrases – consolidating the understanding of “if and only if (necessary and sufficient) condition”, “implies”, “and/or”, “implied” by”, “and”, “or”, “there exists” and through variety of examples related to real life and Mathematics. Validating the statements involving the connecting words – difference between contradiction, converse and contrapositive. Mathematical induction
- Linear Inequalities, solution of linear inequalities in one variable (Algebraic) and two variables (Graphical)
|
| Trigonometry |
- Measurement of angles in radians and degrees, positive and negative angles, trigonometric ratios, functions with their graphs and identities.
- Solution of trigonometric equations.
- Inverse trigonometric functions
|
| Two-dimensional Coordinate Geometry |
- Cartesian coordinates, distance between two points, section formulae, shift of origin.
- Straight lines and pair of straight lines: Equation of straight lines in various forms, angle between two lines, distance of a point from a line, lines through the point of intersection of two given lines, equation of the bisector of the angle between two lines, concurrent lines.
- Circles: Equation of circle in standard form, parametric equations of a circle.
- Conic sections: parabola, ellipse and hyperbola their eccentricity, directrices & foci.
|
| Three dimensional Coordinate Geometry |
- Coordinate axes and coordinate planes, distance between two points, section formula, direction cosines and direction ratios, equation of a straight line in space and skew lines.
- Angle between two lines whose direction ratios are given, shortest distance between two lines.
- Equation of a plane, distance of a point from a plane, condition for coplanarity of three lines, angles between two planes, angle between a line and a plane.
|
| Differential calculus |
- Domain and range of a real valued function, Limits and Continuity of the sum, difference, product and quotient of two functions, Differentiability.
- Derivative of different types of functions (polynomial, rational, trigonometric, inverse trigonometric, exponential, logarithmic, implicit functions), derivative of the sum, difference, product and quotient of two functions, chain rule, parametric form.
- Geometric interpretation of derivative, Tangents and Normal.
- Increasing and decreasing functions, Maxima and minima of a function.
- Rolle’s Theorem, Mean Value Theorem and Intermediate Value Theorem.
|
| Integral calculus |
- Integration as the inverse process of differentiation, indefinite integrals of standard functions.
- Methods of integration: Integration by substitution, Integration by parts, integration by partial fractions, and integration by trigonometric identities.
- Definite integrals and their properties, Fundamental Theorem of Integral Calculus, applications in finding areas under simple curves.
- Application of definite integrals to the determination of areas of regions bounded by simple curves.
|
| Ordinary Differential Equations |
- Order and degree of a differential equation, formulation of a differential equation whose general solution is given, variables separable method.
- Solution of homogeneous differential equations of first order and first degree
- Linear first order differential equations
|
| Probability |
- Various terminology in probability, axiomatic and other approaches of probability, addition and multiplication rules of probability.
- Conditional probability, total probability and Bayes theorem
- Independent events
- Discrete random variables and distributions with mean and variance.
|
| Vectors |
- Direction ratio/cosines of vectors, addition of vectors, scalar multiplication, and position vector of a point dividing a line segment in a given ratio.
- Dot and cross products of two vectors, projection of a vector on a line.
- Scalar triple products and their geometrical interpretations.
|
| Statistics |
- Measures of dispersion
- Analysis of frequency distributions with equal means but different variances
|
| Linear Programming |
- Various terminology and formulation of linear Programming
- Solution of linear Programming using graphical method, feasible and infeasible regions, feasible and infeasible solutions, optimal feasible solutions (upto three non trivial constraints)
|
| Mathematical modeling |
Formulation of simple real life problems, solutions using matrices, calculus and linear programming. |
BITSAT 2024 English Proficiency Syllabus
The following is the BITSAT 2024 English Proficiency Syllabus:-
| Topics |
| Grammar |
- Agreement, Time and Tense, Parallel construction, Relative pronouns
- Determiners, Prepositions, Modals, Adjectives
- Voice, Transformation
- Question tags, Phrasal verbs
|
| Vocabulary |
- Synonyms, Antonyms, Odd Word, One Word, Jumbled letters, Homophones, Spelling
- Contextual meaning.
- Analogy
|
| Reading Comprehension |
- Content/ideas
- Vocabulary
- Referents
- Idioms/Phrases
- Reconstruction (rewording)
|
| Composition |
- Rearrangement
- Paragraph Unity
- Linkers/Connectives
|
BITSAT 2024 Logical Reasoning Syllabus
The following is the BITSAT 2024 Logical Reasoning Syllabus:-
| Topics |
| Verbal Reasoning |
- Analogy: Analogy means correspondence. In the questions based on analogy, a particular relationship is given and another similar relationship has to be identified from the alternatives provided.
- Classification: Classification means to assort the items of a given group on the basis of certain common qualities they possess and then spot the odd option out.
- Series Completion: Here a series of numbers or letters are given and one is asked to either complete the series or find out the wrong part in the series.
- Logical Deduction: Reading Passage: Here a brief passage is given and based on the passage the candidate is required to identify the correct or incorrect logical conclusions.
- Chart Logic: Here a chart or a table is given that is partially filled in and asks to complete it in accordance with the information given either in the chart/table or in the question.
|
| Nonverbal Reasoning |
- Pattern Perception: Here a certain pattern is given and generally a quarter is left blank. The candidate is required to identify the correct quarter from the given four alternatives.
- Figure Formation and Analysis: The candidate is required to analyze and form a figure from various given parts.
- Paper Cutting: It involves the analysis of a pattern that is formed when a folded piece of paper is cut into a definite design.
- Figure Matrix: In this, more than one set of figures is given in the form of a matrix, all of them following the same rule. The candidate is required to follow the rule and identify the missing figure.
- Rule Detection: Here a particular rule is given and it is required to select from the given sets of figures, a set of figures, that obeys the rule and forms the correct series.
|
BITSAT 2024 Biology Syllabus
The following is the BITSAT 2024 Biology Syllabus:-
| Topics |
| Diversity in Living World |
- Biology – its meaning and relevance to mankind
- What is living; Taxonomic categories and aids; Systematics and Binomial system of nomenclature.
- Introductory classification of living organisms (Two-kingdom system, Five-kingdom system);
- Plant kingdom – Salient features of major groups (Algae to Angiosperms);
- Animal kingdom – Salient features of Nonchordates up to phylum, and Chordates up to class level.
|
| Cell: The Unit of Life; Structure and Function |
- Cell wall; Cell membrane; Endomembrane system (ER, Golgi apparatus/Dictyosome, Lysosomes, Vacuoles); Mitochondria; Plastids; Ribosomes; Cytoskeleton; Cilia and Flagella; Centrosome and Centriole; Nucleus; Microbodies.
- Structural differences between prokaryotic and eukaryotic, and between plant and animal cells.
- Cell cycle (various phases); Mitosis; Meiosis.
- Biomolecules – Structure and function of Carbohydrates, Proteins, Lipids, and Nucleic acids.
- Enzymes – Chemical nature, types, properties and mechanism of action.
|
| Genetics and Evolution |
- Mendelian inheritance; Chromosome theory of inheritance; Gene interaction; Incomplete dominance; Codominance; Complementary genes; Multiple alleles;
- Linkage and Crossing over; Inheritance patterns of hemophilia and blood groups in humans.
- DNA –its organization and replication; Transcription and Translation;
- Gene expression and regulation; DNA fingerprinting.
- Theories and evidence of evolution, including modern Darwinism.
|
| Structure and Function – Plants |
- Morphology of a flowering plant; Tissues and tissue systems in plants; Anatomy and function of root, stem (including modifications), leaf, inflorescence, flower (including position and arrangement of different whorls, placentation), fruit and seed; Types of fruit; Secondary growth;
- Absorption and movement of water (including diffusion, osmosis and water relations of cell) and of nutrients; Translocation of food; Transpiration and gaseous exchange; Mechanism of stomatal movement.
- Mineral nutrition – Macro- and micronutrients in plants including deficiency disorders; Biological nitrogen fixation mechanism.
- Photosynthesis – Light reaction, cyclic and non-cyclic photophosphorylation; various pathways of carbon dioxide fixation; Photorespiration; Limiting factors.
- Respiration – Anaerobic, Fermentation, Aerobic; Glycolysis, TCA cycle; Electron transport system; Energy relations.
|
| Structure and Function – Animals |
- Human Physiology – Digestive system – organs, digestion and absorption; Respiratory system – organs, breathing and exchange and transport of gasses.
- Body fluids and circulation – Blood, lymph, double circulation, regulation of cardiac activity; Hypertension, Coronary artery diseases.
- Excretion system – Urine formation, regulation of kidney function
- Locomotion and movement – Skeletal system, joints, muscles, types of movement.
- Control and coordination – Central and peripheral nervous systems, structure and function of neuron, reflex action and sensory reception; Role of various types of endocrine glands; Mechanism of hormone action.
|
| Reproduction, Growth and Movement in Plants |
- Asexual methods of reproduction;
- Sexual Reproduction – Development of male and female gametophytes; Pollination (Types and agents); Fertilization; Development of embryo, endosperm, seed and fruit (including parthenocarpy and helminth).
- Growth and Movement – Growth phases; Types of growth regulators and their role in seed dormancy, germination and movement;
- Apical dominance; Senescence; Abscission; Photo- periodism; Vernalisation;
- Various types of movements.
|
| Reproduction and Development in Humans |
- Male and female reproductive systems;
- Menstrual cycle; Gamete production; Fertilization; Implantation;
- Embryo development;
- Pregnancy and parturition;
- Birth control and contraception.
|
| Ecology and Environment |
- Meaning of ecology, environment, habitat and niche.
- Ecological levels of organization (organism to biosphere); Characteristics of Species, Population, Biotic Community and Ecosystem; Succession and Climax. Ecosystem – Biotic and abiotic components; Ecological pyramids; Food chain and Food web;
- Energy flow; Major types of ecosystems including agroecosystem.
- Ecological adaptations – Structural and physiological features in plants and animals of aquatic and desert habitats.
- Biodiversity and Environmental Issues – Meaning, types and conservation strategies (Biosphere reserves, National parks and Sanctuaries), Air and Water Pollution (sources and major pollutants); Global warming and Climate change; Ozone depletion; Noise pollution; Radioactive pollution; Methods of pollution control (including an idea of bioremediation); Deforestation; Extinction of species (Hot Spots).
|
| Biology and Human Welfare |
- Animal husbandry – Livestock, Poultry, Fisheries; Major animal diseases and their control. Pathogens of major communicable diseases of humans caused by fungi, bacteria, viruses, protozoans and helminths, and their control.
- Cancer; AIDS.
- Adolescence and drug/alcohol abuse;
- Basic concepts of immunology.
- Plant Breeding and Tissue Culture in crop improvement
|
| Biotechnology and its Applications |
- Microbes as ideal system for biotechnology;
- Microbial technology in food processing, industrial production (alcohol, acids, enzymes, antibiotics), sewage treatment and energy generation.
- Steps in recombinant DNA technology – restriction enzymes, NA insertion by vectors and other methods, regeneration of recombinants
- Applications of R-DNA technology in human health –Production of Insulin, Vaccines and Growth hormones, Organ transplant, Gene therapy.
- Applications in Industry and Agriculture – Production of expensive enzymes, strain improvement to scale up bioprocesses, GM crops by transfer of genes for nitrogen fixation, herbicide-resistance and pest-resistance including Bt crops.
|
BITSAT 2024 Slot Booking
BITSAT 2024 slot booking is a crucial process that allows candidates to select their preferred exam date and time slot for the BITSAT 2024 exam.
How to book a slot for BITSAT 2024?
The following is how you can book a slot for BITSAT 2024:-
- Go to the official BITSAT 2024 website at https://www.bitsadmission.com/index.aspx.
- Find and click on the “Slot Booking” link.
- This action will open a new window known as the ‘Online Test Booking System (OTBS).’
- Log in to the OTBS using your BITSAT 2024 application number and password.
- Choose your preferred BITSAT 2024 test center from the list of available options.
- Select your desired test date and the time slot for BITSAT 2024.
- Once the BITSAT slot booking for 2024 is completed, a confirmation message will appear on the screen.
BITSAT 2024 Admit Card
The BITSAT 2024 Admit Card has not yet been released and is expected to be released in May 2024 and June 2024 for Session- I and II, respectively.
The BITSAT 2024 admit card is an essential document that candidates must carry to the examination center.
How to download BITSAT 2024 hall ticket?
The following is how you can download the BITSAT 2024 hall ticket:-
- Open the BITS admission portal.
- Log in by providing your application number and password.
- Locate and click on the “BITS Admission Test Ticket” tab.
- Your BITSAT 2024 admit card will appear on the screen.
- Download the BITS hall ticket and print a hard copy for reference.

In case a candidate finds any error or mistake in the admit card of BITSAT 2024, he or she should contact the authorities immediately at:-
Admissions Office
BITS, Pilani
Rajasthan, India
Pin – 333 031
Tel: 01596-242205, 255294, 255330
BITSAT 2024 Admit Card- Details Mentioned
The following details should be mentioned in the BITSAT 2024 Admit Card for it to be considered valid:-
- Candidate’s name.
- Test center name.
- Tes date and time.
- Test center address.
- Candidate’s signature and photograph.
- Exam instructions for BITSAT 2024.
BITSAT 2024 Preparation Tips
- Understand the Syllabus: Begin by thoroughly understanding the BITSAT 2024 syllabus. It is mainly based on the NCERT curriculum for 11th and 12th grades. Having a clear syllabus outline will help you focus your studies.
- Time Management: Create a study schedule that allocates sufficient time for all subjects. Allocate more time to your weaker areas but don’t neglect any subject.
- Quality Study Material: Use the right study materials, including NCERT textbooks and BITSAT 2024 preparation books. Online resources and test series can also be valuable.
- Mock Tests: Regularly take BITSAT 2024 mock tests. These simulate the actual exam environment and help you become familiar with the exam pattern and time constraints.
- Revision: Regularly review and revise what you’ve learned. This is crucial to retaining information and reinforcing your knowledge.
- Problem-Solving Practice: BITSAT 2024 has a strong focus on problem-solving. Practice solving a variety of problems from each subject to improve your analytical skills.
- English and Logical Reasoning: Don’t underestimate the importance of the English and Logical Reasoning section. Read English newspapers, novels, and practice logical reasoning questions to improve in this area.
- Stay Healthy: Maintain a healthy lifestyle. Get adequate sleep, eat well, and exercise regularly. This will help you stay focused and energized during your preparation.
- Time Management During the Exam: In the BITSAT 2024 exam, time management is crucial. Don’t get stuck on difficult questions; move on and come back to them if time allows.
- Stay Calm: On the exam day, stay calm and composed. Nervousness can negatively affect your performance.
BITSAT 2024 Mock Test
BITS Pilani, the conducting body of BITSAT 2024 , provides official sample tests and mock tests on their official website to help students prepare for the BITSAT exam. These official mock tests are a valuable resource because they closely simulate the actual BITSAT exam environment and provide an opportunity to practice under exam-like conditions.
To access these official BITSAT 2024 mock tests, you can visit the official BITSAT 2024 website and look for the “Sample Test” or “Mock Test” section. (https://www.bitsadmission.com/bitsat/BITSAT_SAMPLETEST/default.asp)
BITSAT 2024 Result
The BITSAT 2024 Result has not yet been released but is expected to be released in July 2024.
How to check the BITSAT 2024 Result?
- Go to the official BITS Pilani website at bitsadmission.com.
- Locate and click on the link for the BITSAT2024 scorecard.
- Provide your Application number and password.
- Click the “Submit” button.
- Your BITSAT 2024 Result, including the scorecard, will be shown on the screen.
- Download and print your BITSAT 2024 rank card for future reference.
BITSAT 2024 Result- Tie-Breaking Rule
For all applicants participating in BITSAT 2024 and expressing interest in admission, it is mandatory to submit an admission application form along with their 12th-grade marks and preferences for different degree programs across Pilani, Goa, and Hyderabad campuses. A merit list will be compiled by the authorities based on the candidates’ BITSAT 2024 scores. In instances where two or more candidates obtain identical scores in BITSAT 2024, a tie-breaking criteria will be applied.
Firstly, candidates with higher scores in Mathematics/Biology will be prioritized for a higher rank. If a tie still persists, preference will be given to those with superior Physics scores. Should the tie continue, candidates with better Chemistry scores will be ranked higher. If the tie remains unresolved, the marks obtained in Physics, Chemistry, and Biology/Mathematics (as applicable) in the Class 12th board examination will be considered to break the tie.
BITSAT 2024 Cutoff
The BITSAT 2024 Cutoff has not yet been released, and once released will be updated here then. For the ease and better understanding of the candidate we are therefore, providing you with the BITSAT 2023 Cutoff:-
| Name of the Course |
BITS Pilani Cutoff |
BITS Goa Cutoff |
BITS Hyderabad Cutoff |
| BE Chemical |
189 – 191 |
160 – 165 |
160 – 162 |
| BE Civil |
161 – 167 |
– |
150 – 158 |
| BE Electrical & Electronics |
250 – 260 |
230 – 239 |
220 – 230 |
| BE Mechanical |
218 – 228 |
188 – 194 |
178 – 186 |
| BE Manufacturing |
179 – 187 |
– |
– |
| BPharm |
120 – 125 |
– |
99 – 109 |
| BE Computer Science |
300 – 320 |
280 – 286 |
269 – 280 |
| BE Electronics & Instrumentation |
230 – 250 |
220 – 227 |
220 – 228 |
| BE Electronics & Communication |
270 – 279 |
250 – 258 |
250 – 255 |
| MSc Biological Sciences |
167 – 171 |
154 – 165 |
150 – 160 |
| MSc Chemistry |
160 – 168 |
158 – 165 |
155 – 163 |
| MSc Economics |
240 – 249 |
220 – 240 |
200 – 220 |
| MSc Mathematics |
210 – 220 |
180 – 190 |
170 – 180 |
| MSc Physics |
210 – 218 |
180 – 190 |
170 – 176 |
BITSAT 2024 Counseling
BITSAT 2024 counseling is the process through which candidates who have qualified for the BITSAT 2024 are allotted seats in various degree programs offered by BITS campuses. The following are the key steps and information related to BITSAT 2024 counseling:-
- Announcement of Counseling: After the BITSAT 2024 results are declared, the institute will release a notification about the counseling process. This includes the schedule and details for the counseling sessions.
- Filling Admission Application Form: Candidates need to fill an online admission application form where they provide their 12th-grade marks and their preferences for the degree programs and campuses.
- Merit List: BITS prepares a merit list based on BITSAT 2024 scores and 12th-grade marks. The candidates are then sorted based on their preferences and available seats.
- Seat Allotment: During the counseling process, candidates are allotted seats according to their merit and preferences. The highest-ranked candidate gets the first choice, and this process continues until all seats are allocated.
- Document Verification: Candidates must bring their original documents for verification during the counseling process. This includes marksheets, certificates, and other relevant documents.
- Payment of Fees: Once a seat is allotted, candidates need to pay the admission fees to confirm their seat. These fees are non-refundable and non-transferable.
- Reporting to Campus: After the seat is confirmed, candidates must report to the respective BITS campus on the specified date to complete the admission process.
- Waiting List: In case some seats remain vacant after the initial rounds of counseling, a waiting list is generated. Candidates on the waiting list may get a chance if seats become available.
- Additional Iterations: If necessary, additional iterations of counseling may take place to fill any remaining seats.
BITSAT 2024 counseling is highly competitive, and the availability of seats in preferred programs and campuses depends on the candidate’s BITSAT 2024 score, 12th-grade marks, and choices made during the admission application process.Applicants who have completed their application forms must submit a counseling fee as part of the BITSAT 2024 admissions procedure. Candidates are asked to pay the 1000 INR BITSAT 2024 counseling costs using one of the provided payment options. It’s essential for candidates to be well-prepared and have a clear understanding of their preferences to maximize their chances of securing admission to their desired program.
BITSAT 2024 FAQs
Q How can I check my BITSAT 2024 seat allotment result?
Ans Candidates will be able to check it by logging in to the official website of BITSAT 2024 by using a valid application number and password.
Q How will the BITSAT 2024 counseling be conducted?
Ans The counseling of BITSAT 2024 will be conducted in online mode only. Candidates need to pay the required fee during the counseling and seat allotment process.
Q What is the BITSAT 2024 marking scheme?
Ans Three marks will be given for correct answers and one mark is reduced for incorrect responses.
Q When will BITS Pilani commence BITSAT 2024 registration?
Ans BITSAT 2024 registration form has been released from January 15, 2024.
Q What will be the total number of questions in BITSAT 2024?
Ans BITSAT 2024 exam consists of 130 questions.
Q How can I check my BITSAT 2024 application form status?
Ans To check the status of the application form, candidates will have to log in to the BITS admission portal 2024 using the details like email id, mobile number, and date of birth.
Conclusion
BITSAT 2024 is expected to take place in the May 2024 and June 2024 for Session-I and II, respectively. The BITSAT 2024 rigorously evaluates their knowledge in subjects like Physics, Chemistry, and Mathematics, in addition to assessing their English language proficiency and logical reasoning capabilities. Achieving success in BITSAT 2024 opens the doors to quality education and exciting career prospects at BITS Pilani, Goa, and Hyderabad campuses. The post gives the candidates a thorough insight into BITSAT 2024, its eligibility criteria, application process, result among other things.
Also See :

![]() BITSAT 2024 Application Form has been released from January 15, 2024. Check here for application form details.
BITSAT 2024 Application Form has been released from January 15, 2024. Check here for application form details.



