CBSE Class 10 Science Notes of Chapter 4 Carbon and its Compound, Video Explanation, Question Answers
Carbon and its Compounds Class 10 notes- Here is a CBSE Class 10 Science Chapter 4 Carbon and its Compound Notes and NCERT Solutions to Important Question Answers. Given here is the complete explanation of the chapter, along with examples and all the exercises, Questions and Answers are given at the back of the chapter.
Science Class 10
Chapter 4 – Carbon and its Compounds Notes
- organic chemistry
- See Video Explanation of Carbon and its Compound
- fermentation
- Rules for IUPAC names
- introduction to carbon
- aldehyde
- soaps and detergents
- special properties of carbon
- carboxylic acid
- NCERT Book solutions
- hydrocarbons
- ester
Organic Chemistry
Carbon and its Compound – Organic chemistry is that branch of chemistry that deals with the study of carbon and its compounds.
Carbon is so versatile in nature that organic chemistry forms a separate branch of chemistry which deals mainly with carbon and its compounds.
Introduction to Carbon
Carbon is an element with symbol C
Atomic number 6
Electronic configuration -2,4
As suggested by its configuration, it belongs to second period and fourteen group.
Its valence electrons are 4
Its valency is also 4
Carbon and its Compound Video Explanation
Carbon and its Compound Related Links –
Special Properties of Carbon
1. Catenation: It is the self linking property of carbon i.e. it links with its own kind of atoms, giving rise to long chains that are branched or ringed or can be straight.

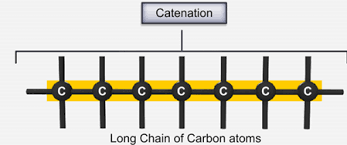
2. Tetravalency: Carbon has 4 electrons in its valence shell that it can easily loose, gain or share with other atoms to attain stability
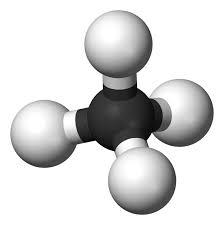
3. Multiple Bonding: It can show multiple bonding. It means that it can form single, double or triple bonds
Due to these special properties carbon is versatile in naturei.e., it forms so many compounds that we have created a special branch to study it.
Firstly, it was concluded that organic compounds can not be made in laboratory as they require a mysterious force to form and that is present only in plants and animals but Fedrich Wohler gave a blow to this conclusion that he accidently prepared the first organic compound that was man made- urea
But the most common organic compound is hydrocarbons that we are going to study in this chapter
Let us study hydrocarbon in detail now :
Hydrocarbons
Hydrocarbons are compounds of carbon that contain hydrogen.
There are two types of hydrocarbons –
- saturated hydrocarbons
- unsaturated hydrocarbons
(Saturated) (Unsaturated)
Alkanes Alkenes Alkynes
single bond double bond triple bond
Ane ene yne
F ? Cn + H2n +2 CnH2n CnH2n-2
n = no. of C atom
Numbers
CH4 C2H4 C2H2
(Methane) (ethane) (elkyne)
C2H6 C3H6 C3H4
(ethane) (Propene) (Propyne)
F. gps:- gp .suffex
Alcohol – OHgp ol
(Hydroxyl)
ketone 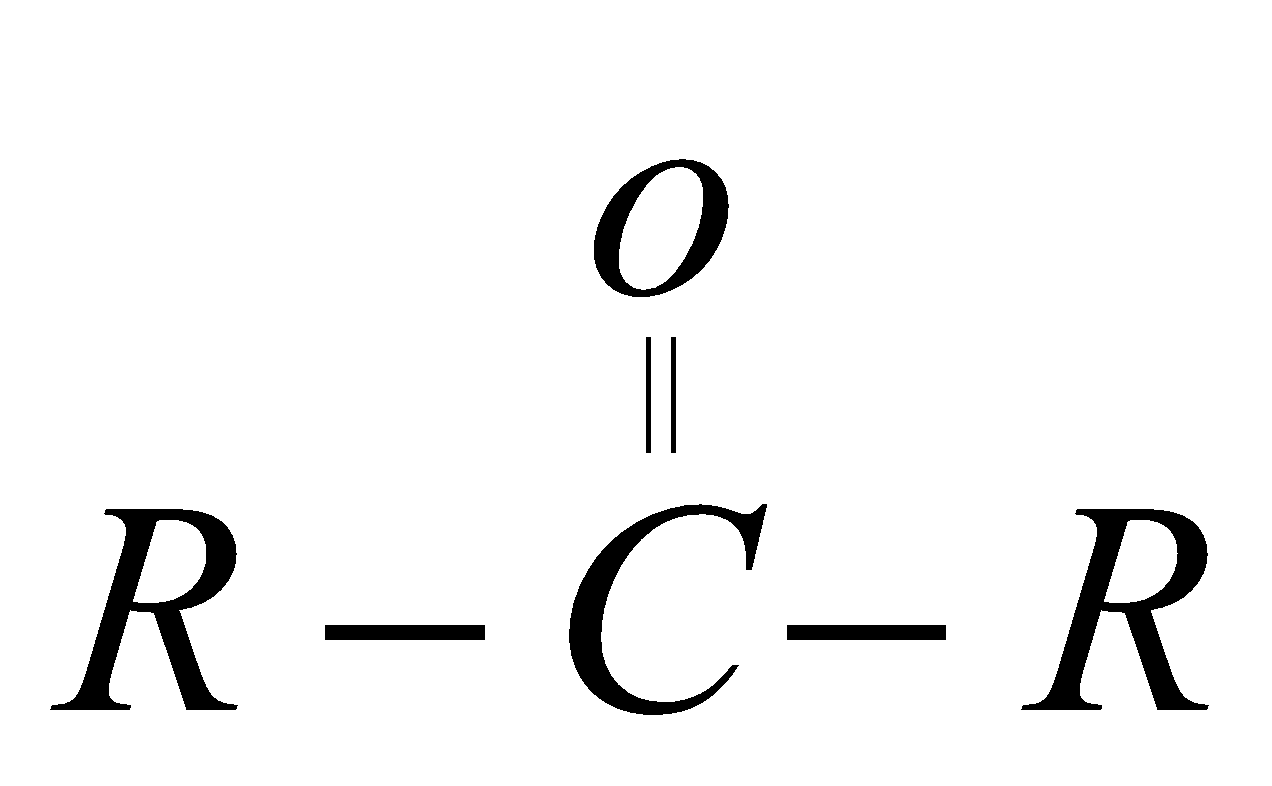 One
One
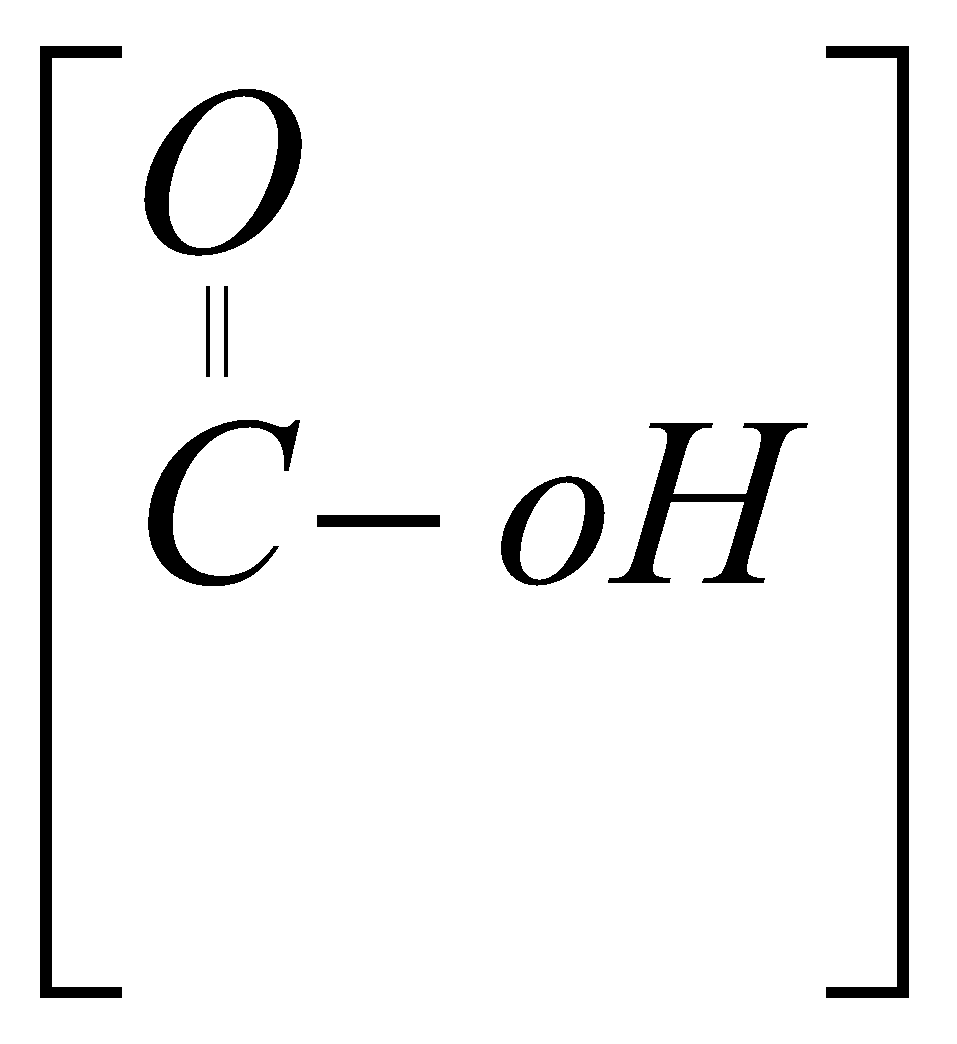
carboxylic acid COOH Oic acid
ester 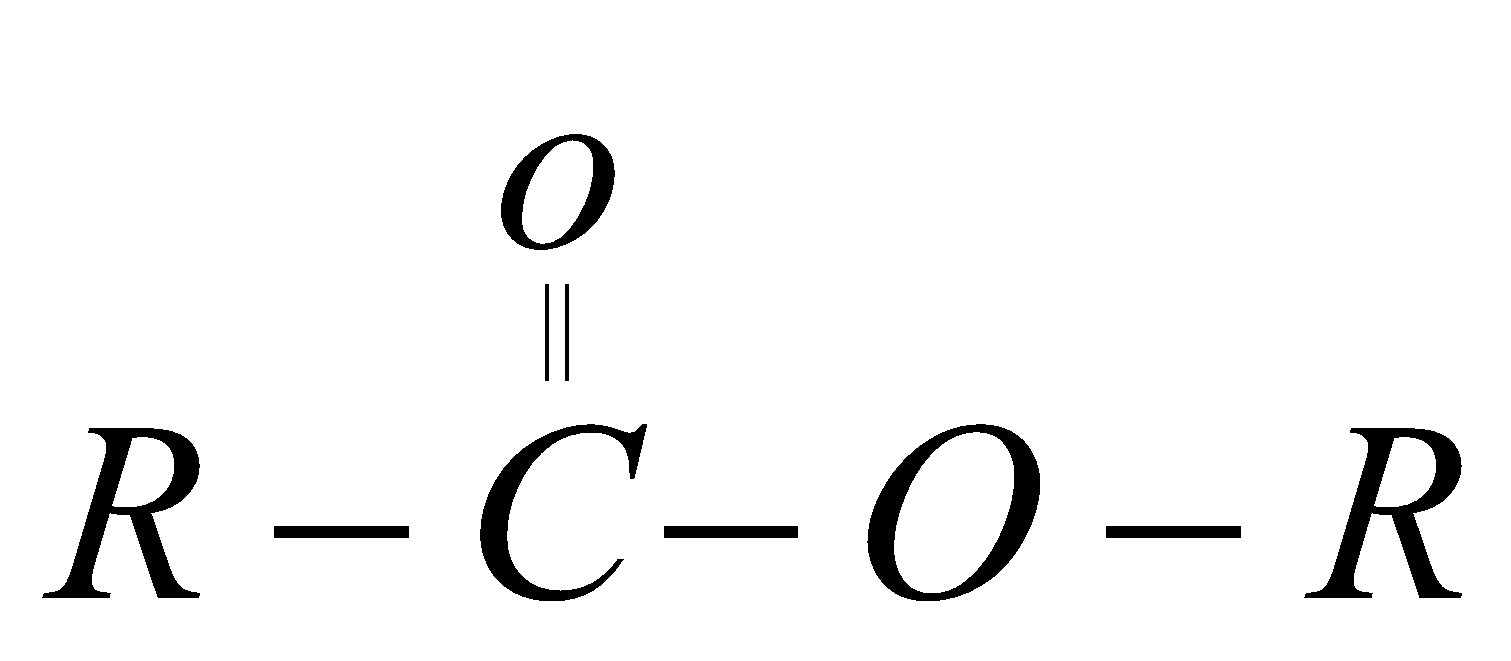 Oate
Oate
R ? Alkyl gp. [ suffex- yl gp.)
for example: – CH3-
(Methyl)
C2H5-
(ethyl)
Alcohol
F.gp ______ OH
Suffex ? ol
Ex ample – CH3OH, C2H5OH
(Methanol) (ethanol)
Related – Tips to score 95% in Class 10 Science Paper
Fermentation
The breakdown of complex substances in to simpler substances by action of enzymes released by anaerobic becteria is called fermentation. By this method we can prepare only ethanol. In this, firstly, sugarcane juice and water are mixed and heated to form sweet liquid called molasses.
Then, in presence of yeast and enzyme invertase, it breaks in to glucose and fructose and then further, glucose and fructose break into further simpler substances called ethanol and carbon dioxide. This method is generally carried out in breweries.
S.juice + H2O ? molasses
C12H22O11 + H2O 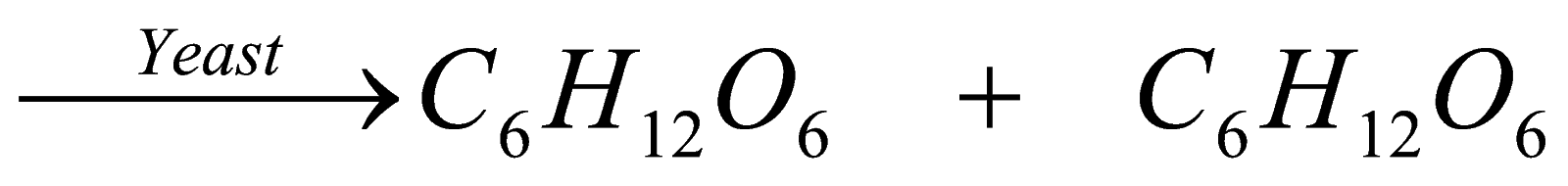
(Sucrose) (invertase ) (glucose) fructose
C6H12O6 C2H5OH + Co2
(zymase) (ethanol)
Hydrolysis of ethane (Hydration of alkene): This is a method by which any alcohol can be prepared and accordingly we take member of alkene.Suppose we need to prepare ethanol than we need to take ethene.
When water is added to ethene in presence of some weak acid like phosphoric acid then ethene undergoes addition reaction and leads to formation of ethanol.
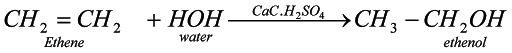 ?
?
By alkyl holide: we can also prepare it by reaction of alkyl halide with aqueous KOH
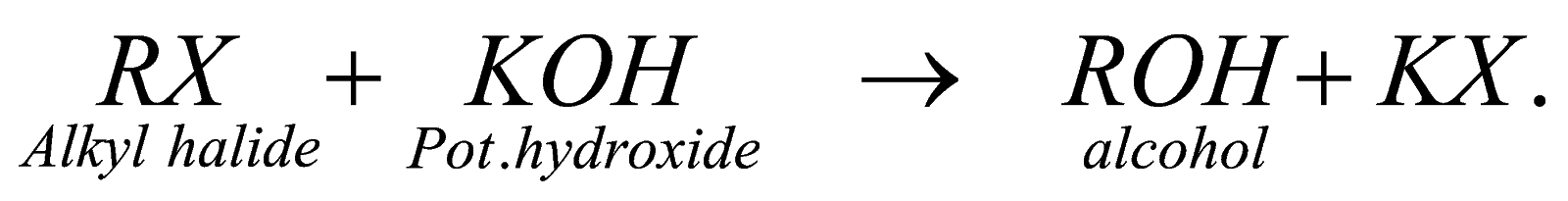
Physical properties
Ethanol
1) lighter than water
2) volatile
3) inflammable
4) Rectified spirit-ethanol mixed with water
Chemical properties
1.oxidation
Alcohol undergoes oxidation in two ways : partial and complete
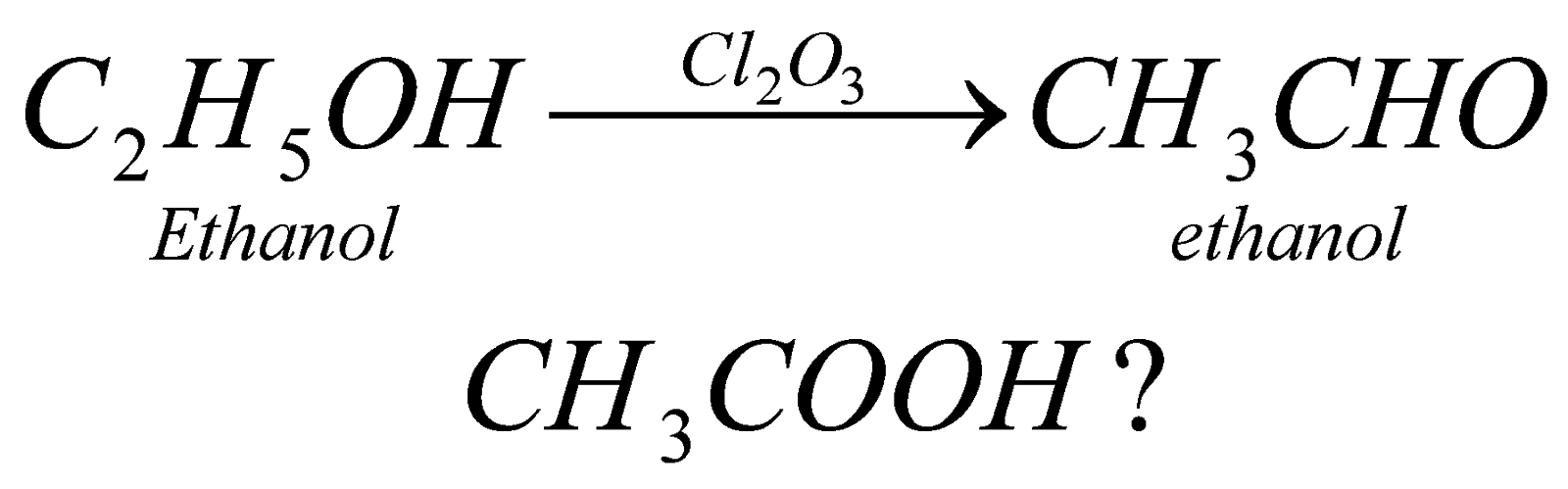
In partial (P.oxide) forms aldehyde . it is neither step down or step up reaction or we can say the number of carbon atoms remains same that is, if we start a reaction with eith member of alcohol, it will form the same eth member of aldehyde. The catalyst however, changes like in case of meth member we take Ag and in case of eth member we take chromic oxide and so on.
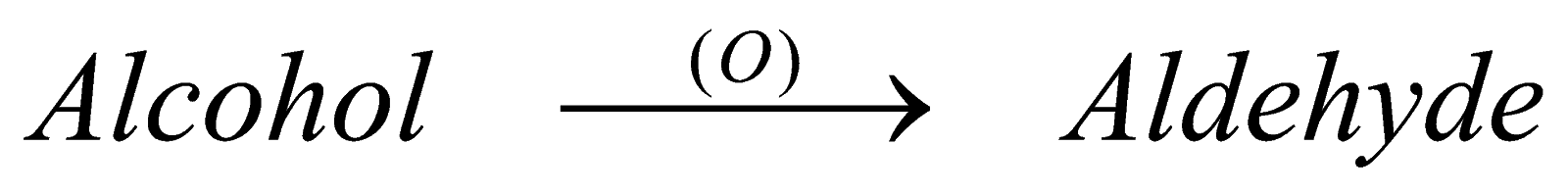
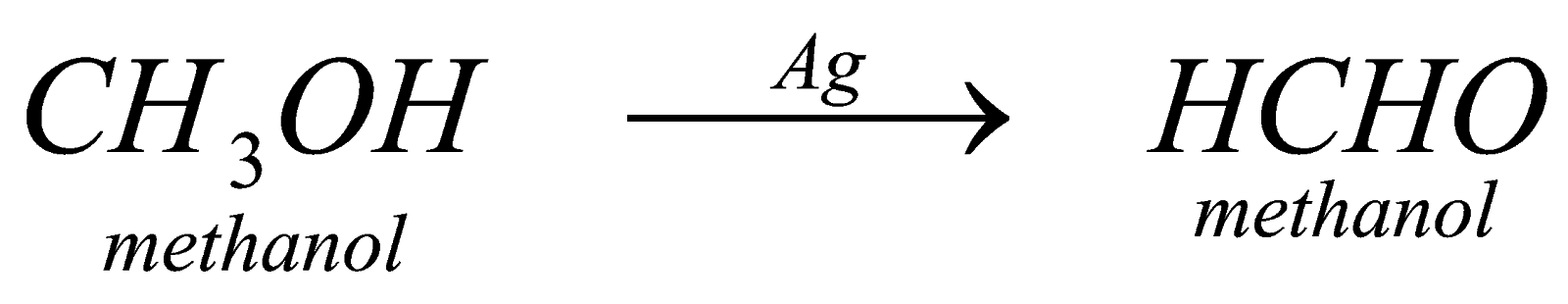
Complete Oxidation: in case of complete oxidation it forms carboxylic acid again with same number of carbon atoms. In this, the catalyst generally we use is a strong oxidising agent – alkaline potassium permagnate or alkaline potassium dichromate
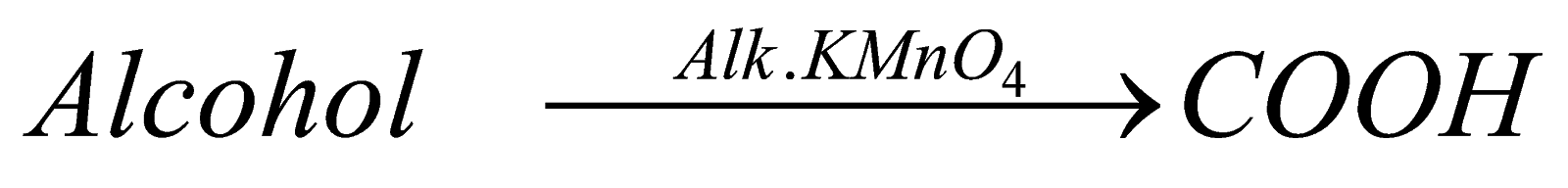

2. Reduction
When we carry on reduction of alcohol it forms alkane in presence of reducing agents like Pt,Ni,Pd, etc. It is also neither step up or step down reaction.
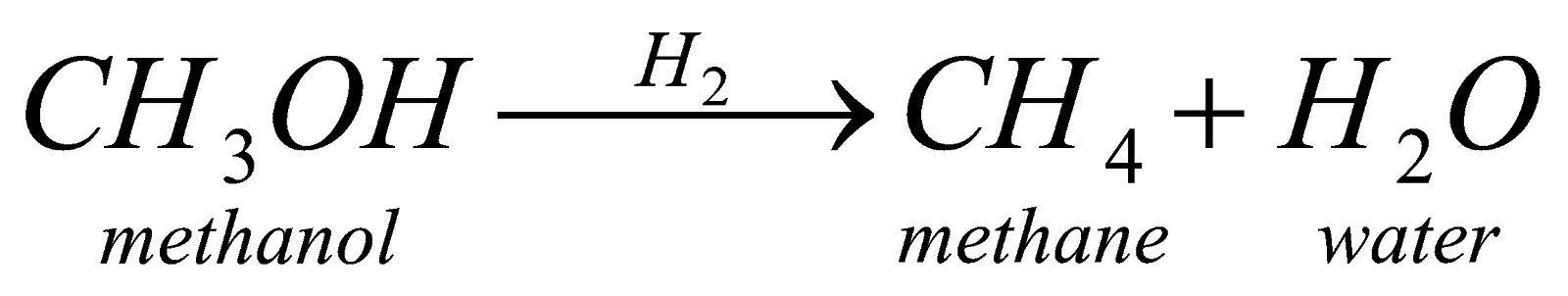
3. Esterification
This is one of the important reaction of alcohol, when it is mixed with carboxylic acid in presence of concentrated sulphuric acid, it forms fruity smelling liquid called ester and this ester is widely used in perfumes , icecreams, etc.
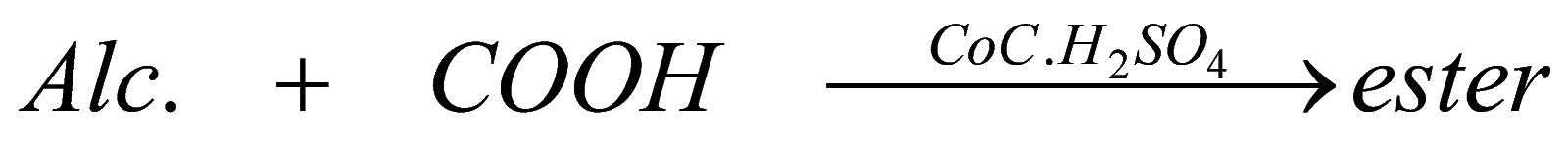
![]()
4. Reaction with metal
When alcohol reacts with any metal that is reactive than hydrogen, it forms metal alkoxide due to displacement of H by metal.
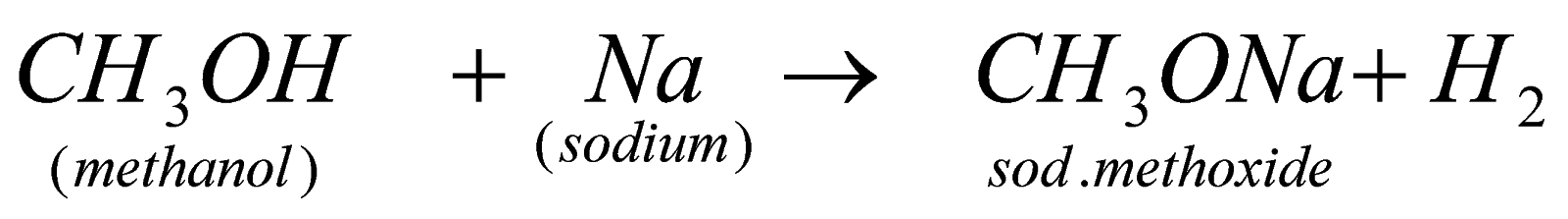
5. combustion
Whenever any carbon compound or its derivative is heated in presence of oxygen it forms carbon dioxide, water vapour and respective gases if some other atom is also present as shown :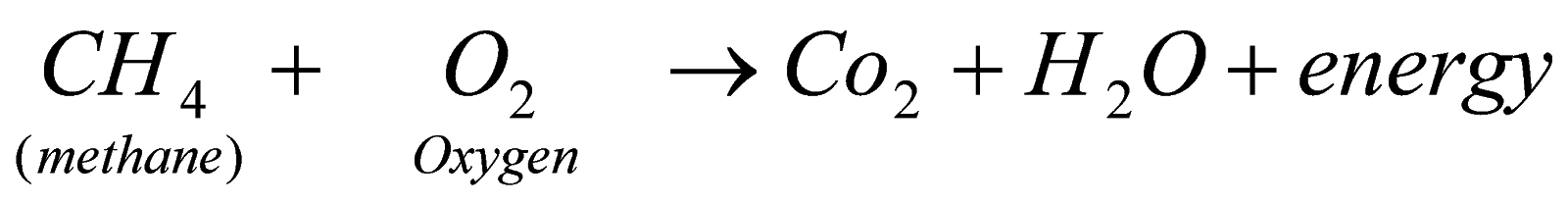
As we have noticed that alcohol is one of the major components of hard drinks and is also used as industrial solvent in industries, it is denatured that is :
Denatured alcohol: is to alter the nature of alcohol by adding poisonous substances like methanol, pyridine, etc. in order to prevent its mis-use in industries by workers.
Class 10 Science Chapters
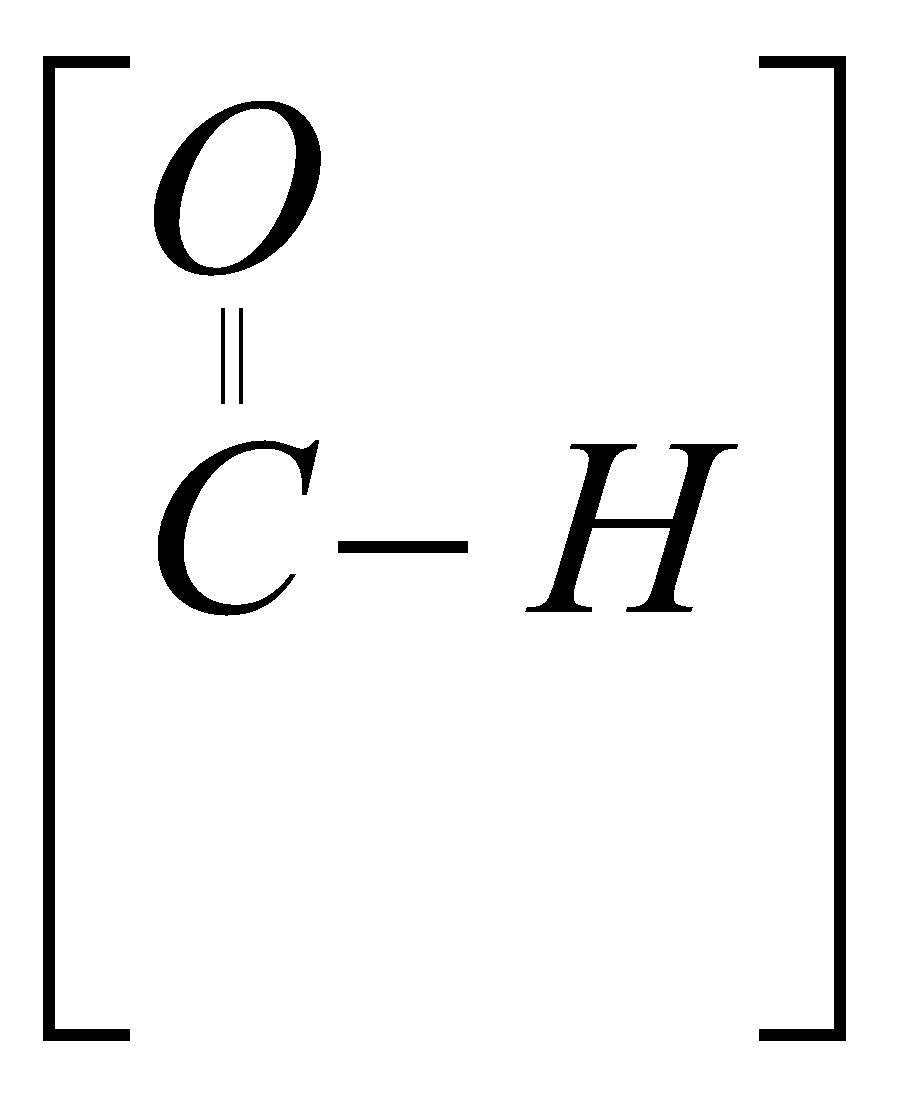
Aldehyde
Gp – CHO
Sfex – al
P :- HCHO CH3CHO �
Methanal ethanal
(formaldehyde) (acetaldehyde)
Preparation-
(i) Oxide of Alcohol: by this method we can prepare any aldehyde. We have to proceed in this reaction by using catalyst depending upon the member of alcohol that we are using like for methanol we will use catalyst silver, etc.
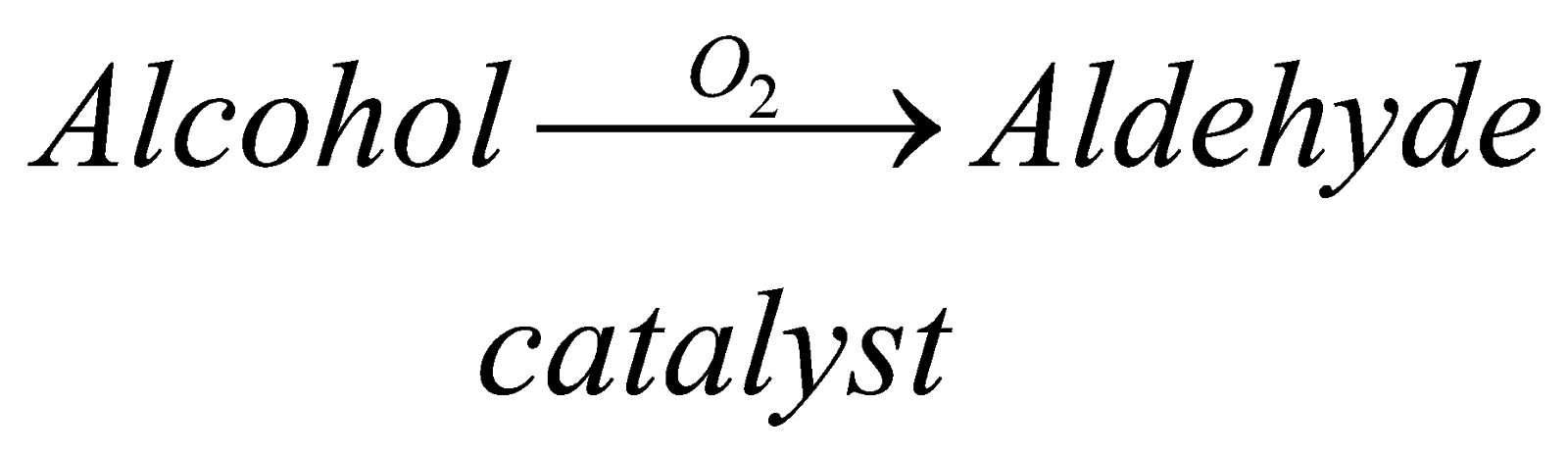
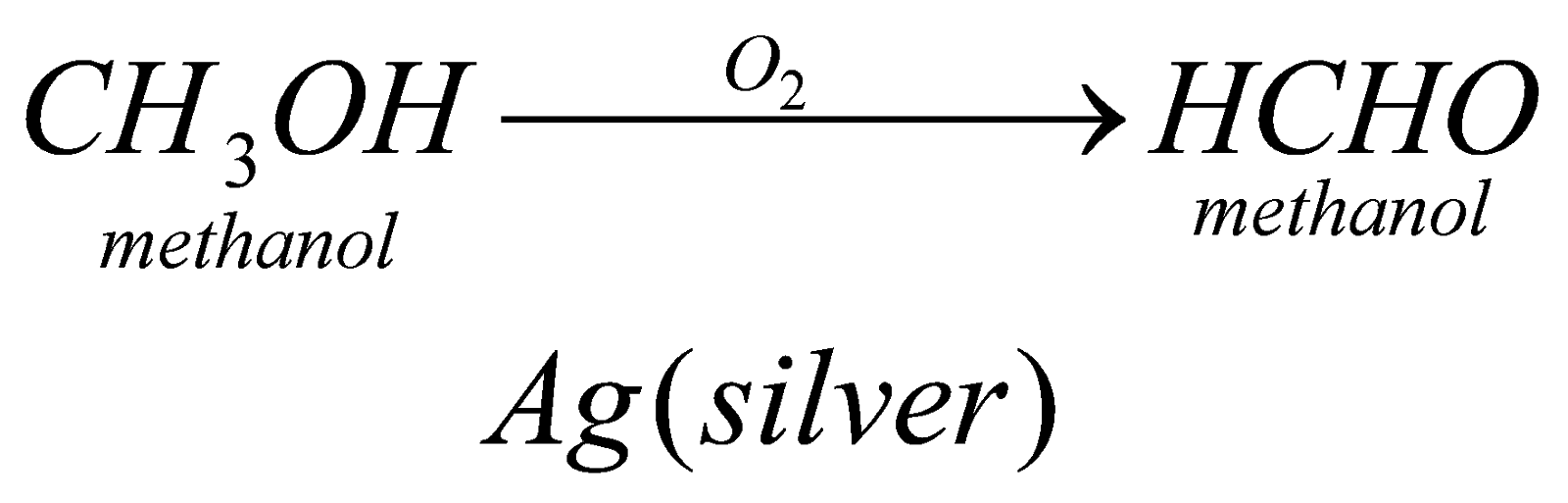
Properties of aldehyde-
1.Oxidation: when aldehyde reacts with oxygen in controlled condition it in the presence of oxidizing agent, it forms respective carboxylic acid. For example, if we oxidize methanol in presence of alkaline potassium permagnate, it forms methanoic acid.
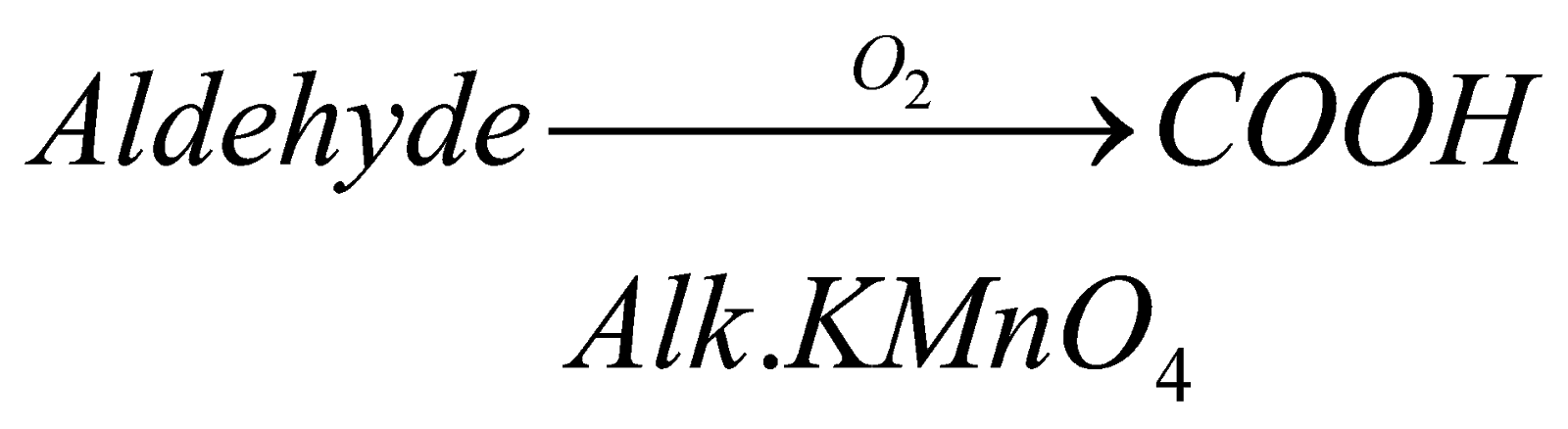
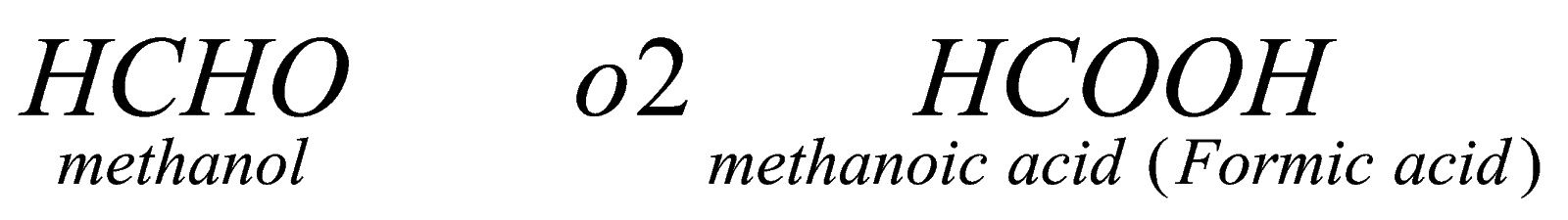
2. Reduction:
When we carry out reducyion of aldehyde that is when we react it with hydrogen in presence of reducing agent, it forms alcohol. For example : when methanal is reduced in presence of nickel, it forms methanol.
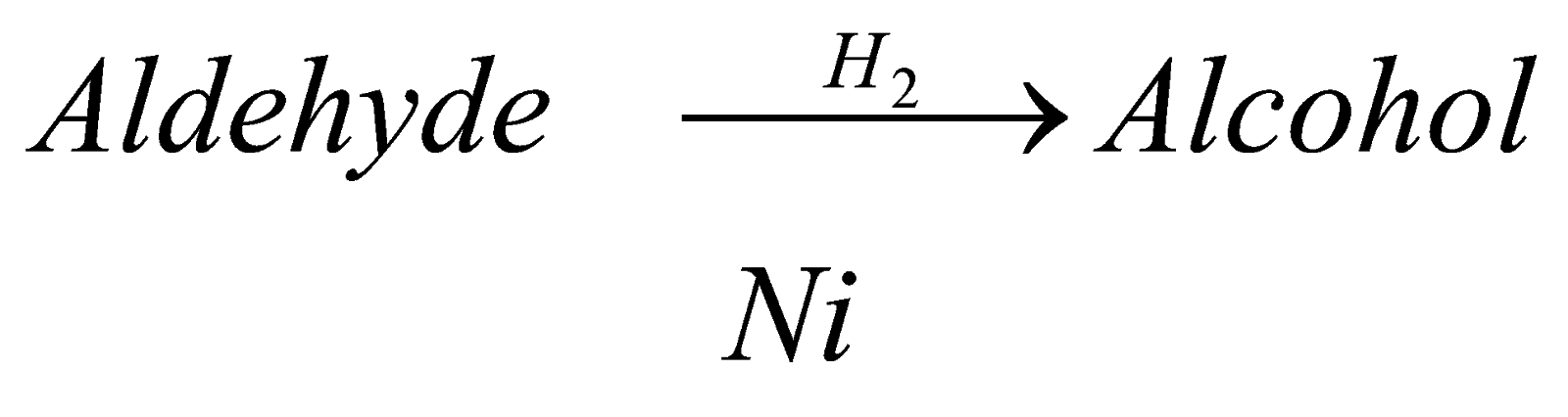
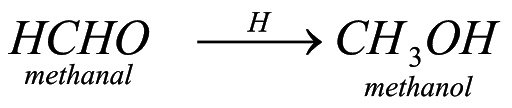
3.Addition reaction:
The aldehyde has double bond character so when any substance is added to it, it breaks its extra bond and adds the substance to it. Like if we add HCN� then H gets attached to oxygen and CN also gets attached to carbon forming cyanohydrins as shown in the reaction.
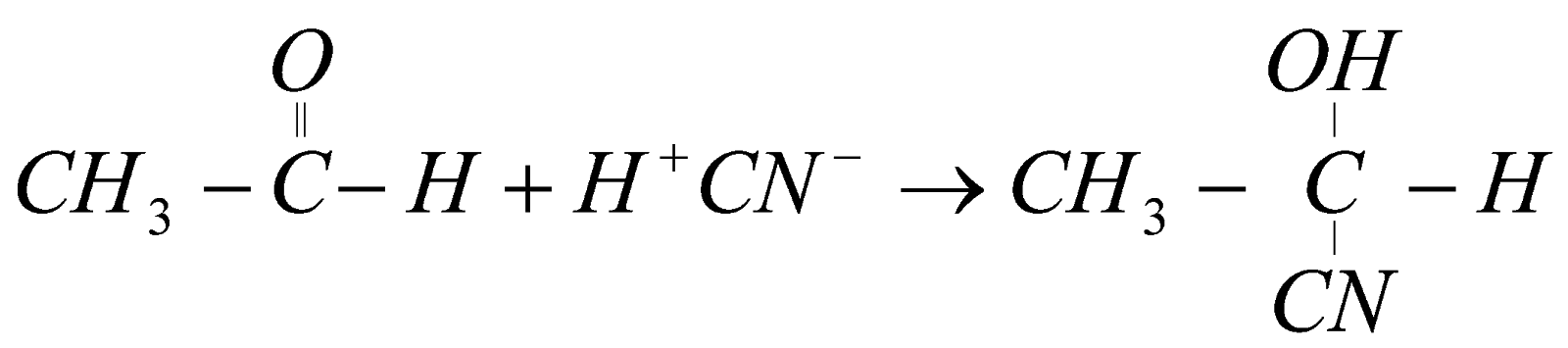
Carboxylic Acid
Fgp. ? COOH
Suffer ? Oic acid

(Formic acid) (Acetic acid)
Preparation –
(i) by Oxidation of Alcohol: The carboxylic acids can be formed by complete oxidation of alcohols in presence of alkaline potassium permagnate.
For example: when we carry out complete oxidation of methanol in presence of strong oxidising agent, it forms methanoic acid.
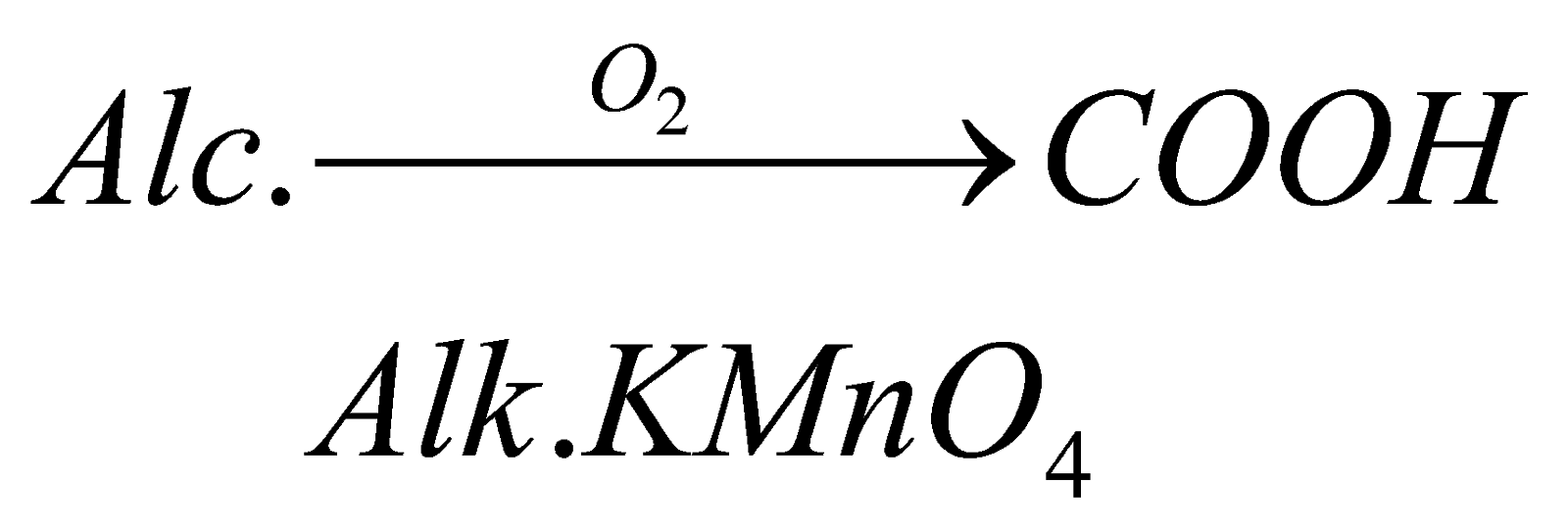
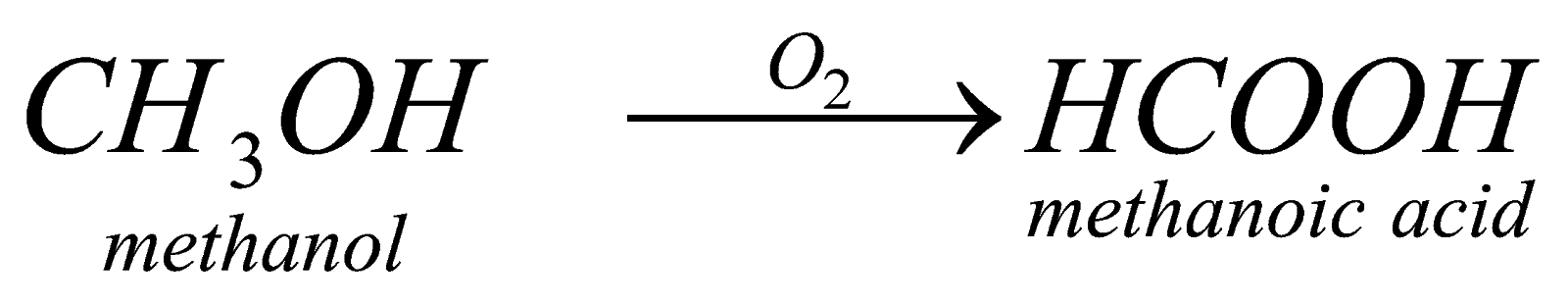
(ii) Oxidation of Aldehyde: it can also be formed by oxidation of aldehyde in presence of strong oxidizing agents .for example in given example methanal gets oxidized to methanoic acid
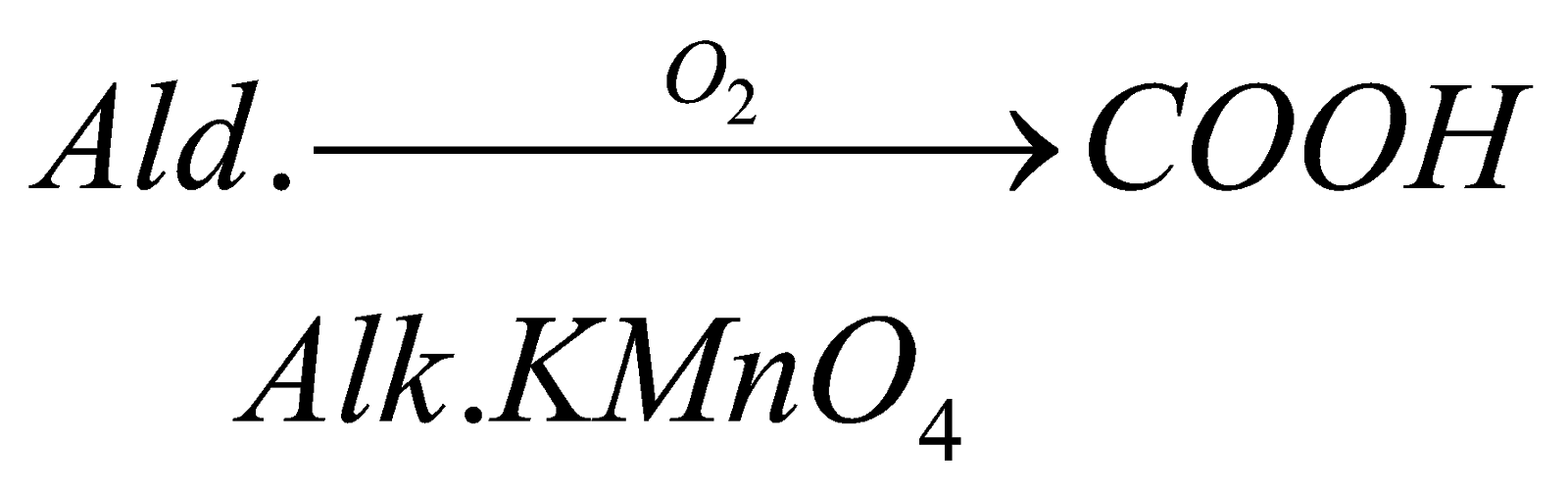
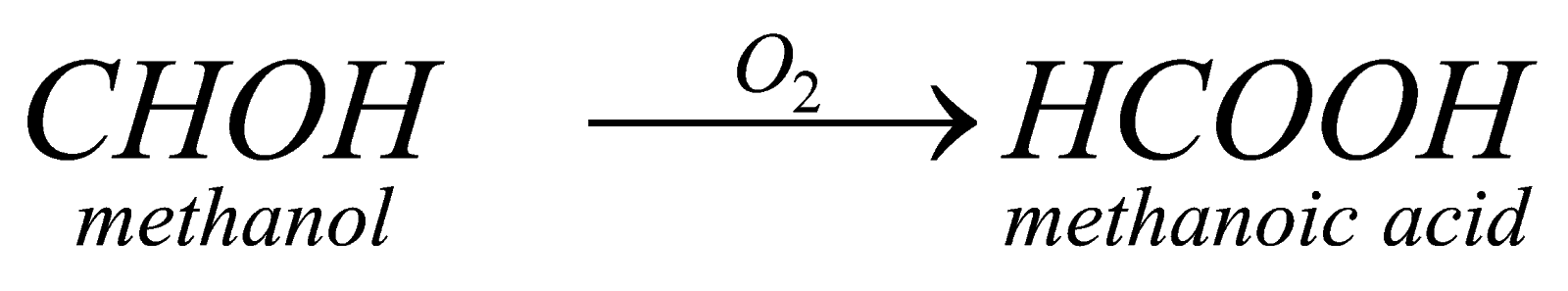
Properties
1. Reduction : when reduction of carboxylic acid is carried out, it forms respective aldehyde. For example in case of reduction of methanoic acid it forms methanal when reduction is carried out in presence of reducing agent like palladium, nickel, etc.
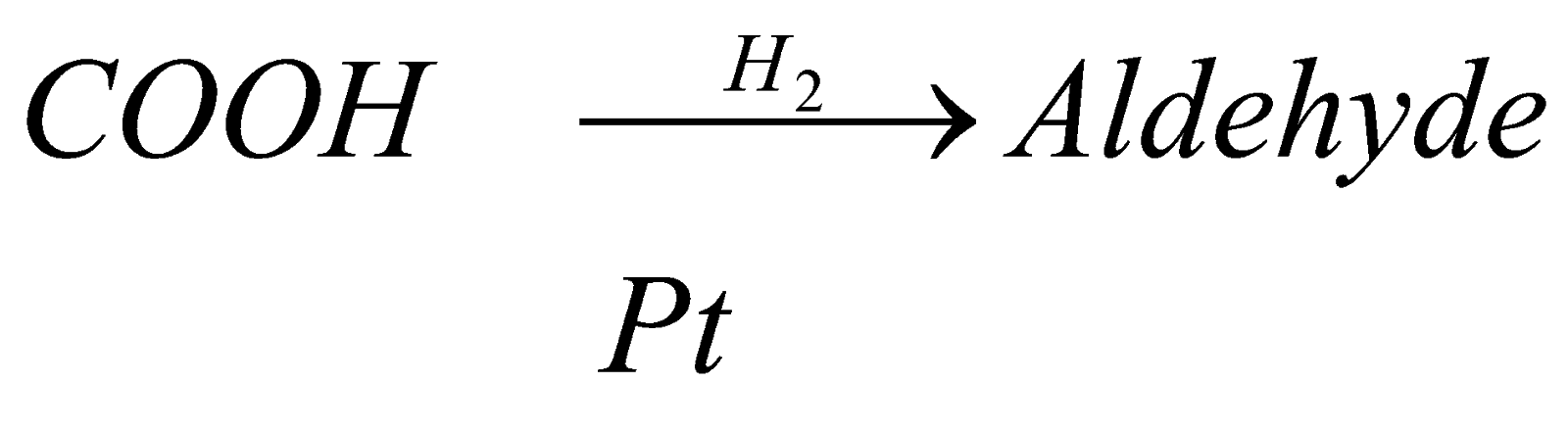
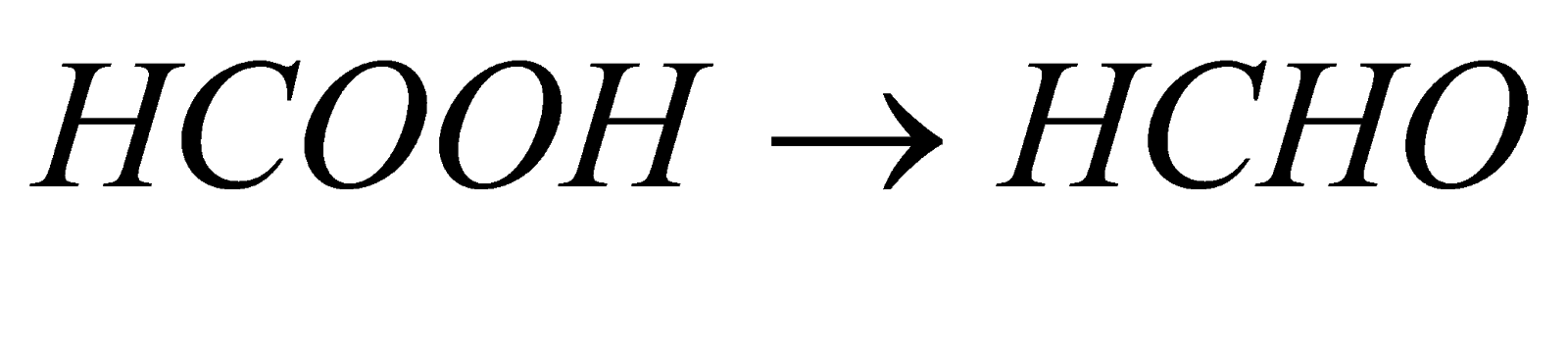
(methanoic acid) (methanal)
2. Reaction with alcohol: when any carboxylic acid reacts with alcohol in presence of concentrated sulphuric acid, it forms ester (a fruity substance liquid).
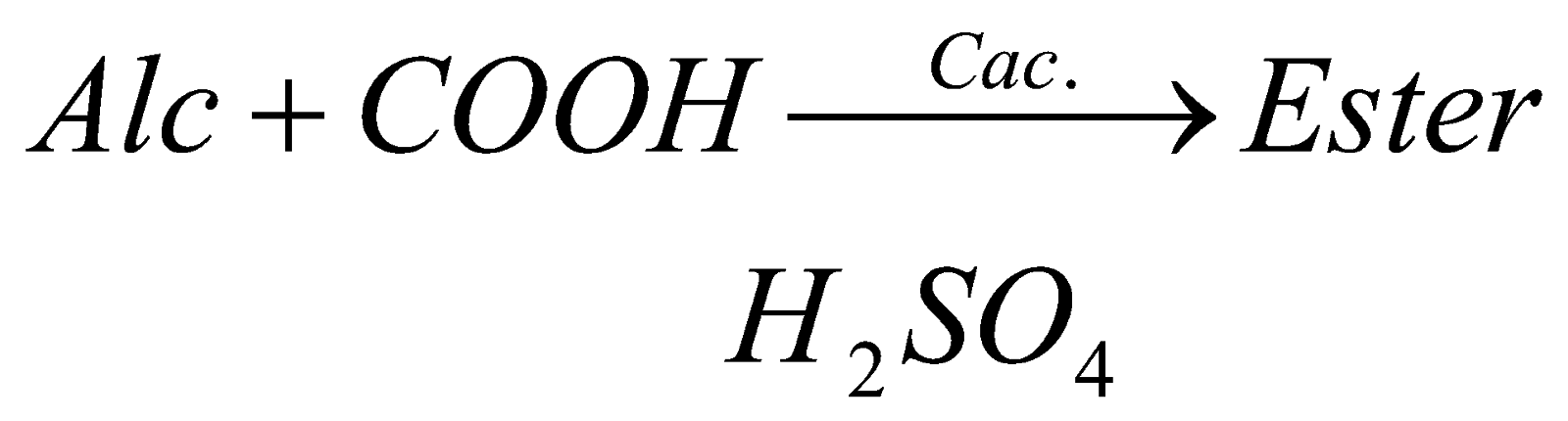
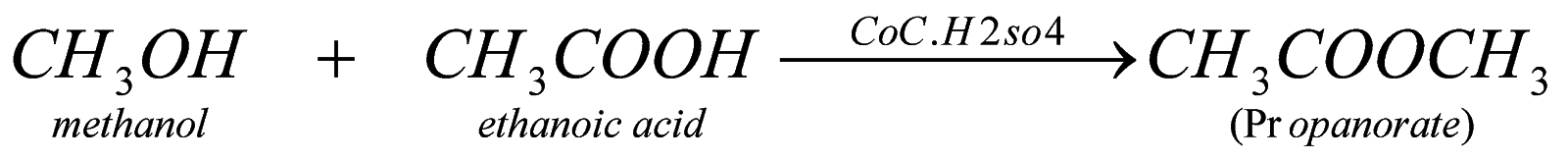 ???
???
3. Reaction with metal: when carboxylic acid reacts with metal, then metal, being more reactive, displaces hydrogen and forms sodium alkanoate.

4. Rxn with metal hydroxide & metal carbonates : when carboxylic acid reacts with metal , then metal being more reactive displaces hydrogen aand form sodium alkanoate and also water and carbondioxide
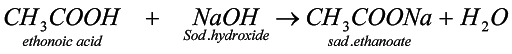
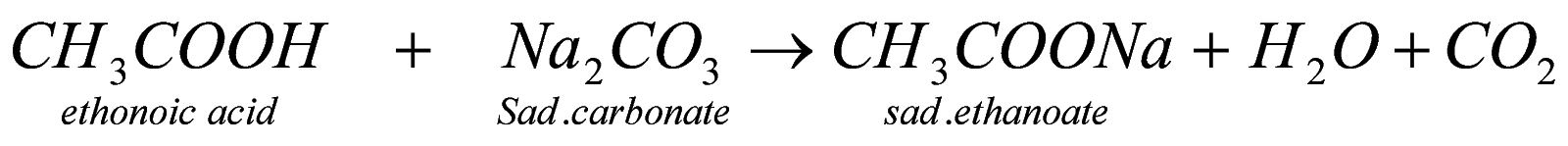
5.Decarboxylation removal by 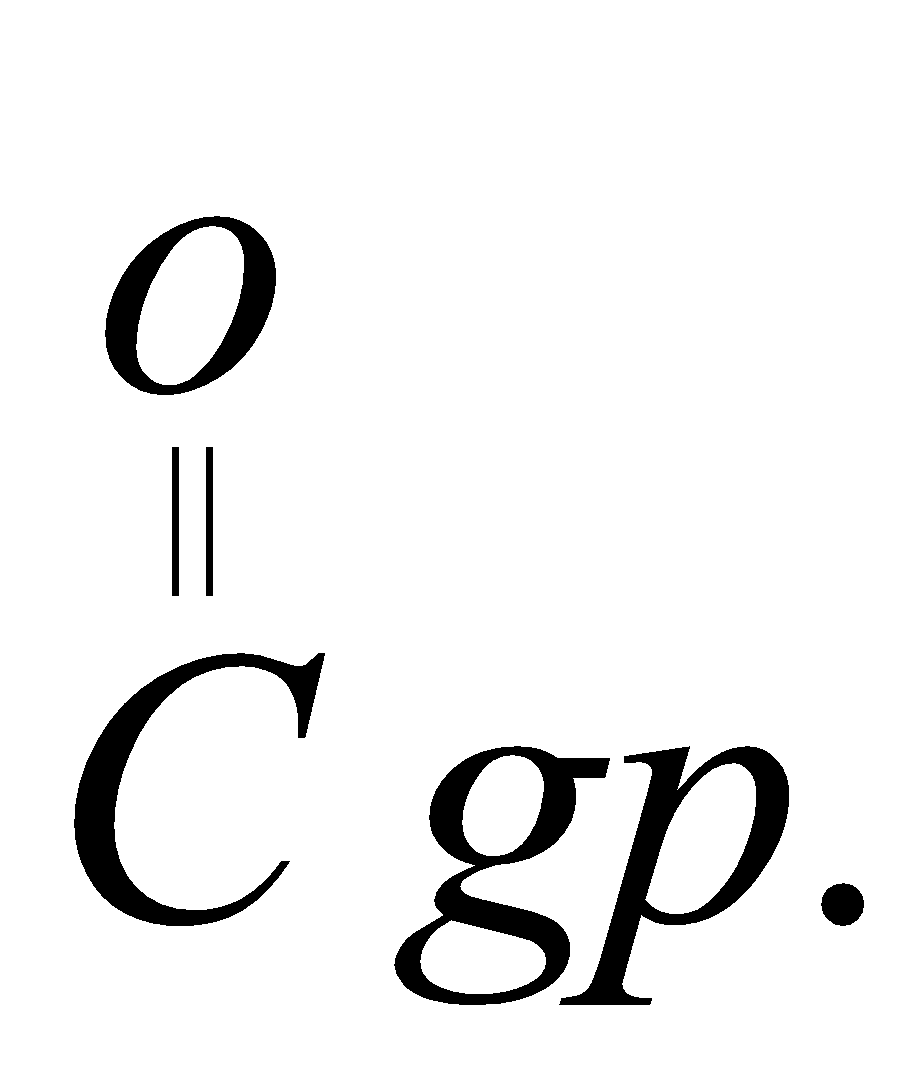 : This reaction involves removal of c=o group and leads to the formation of alkane. This reaction is step down reaction as the main product formed has one carbon less than the parent reactant. Kike in the given example, we started with eth member but the product formed has one carbon. So, it is step down and removal of c=o gp reaction.
: This reaction involves removal of c=o group and leads to the formation of alkane. This reaction is step down reaction as the main product formed has one carbon less than the parent reactant. Kike in the given example, we started with eth member but the product formed has one carbon. So, it is step down and removal of c=o gp reaction.
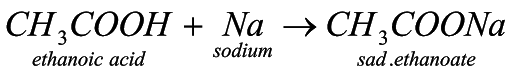
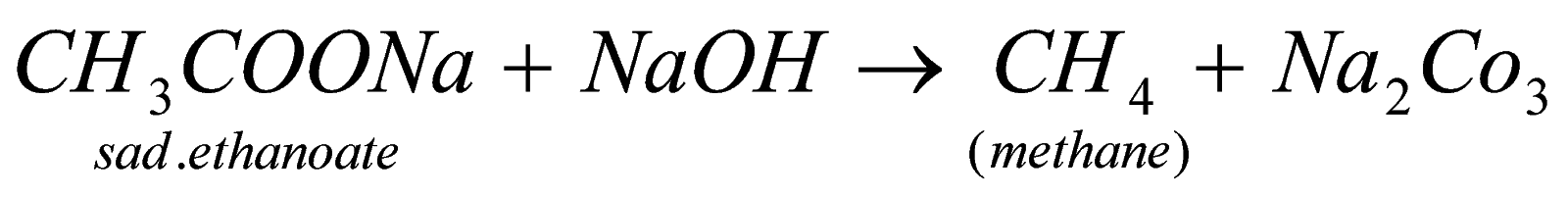
Ester
Preparation
when any carboxylic acid reacts with alcohol in presence of concentrated sulphuric acid it forms ester (a fruity substance liquid)
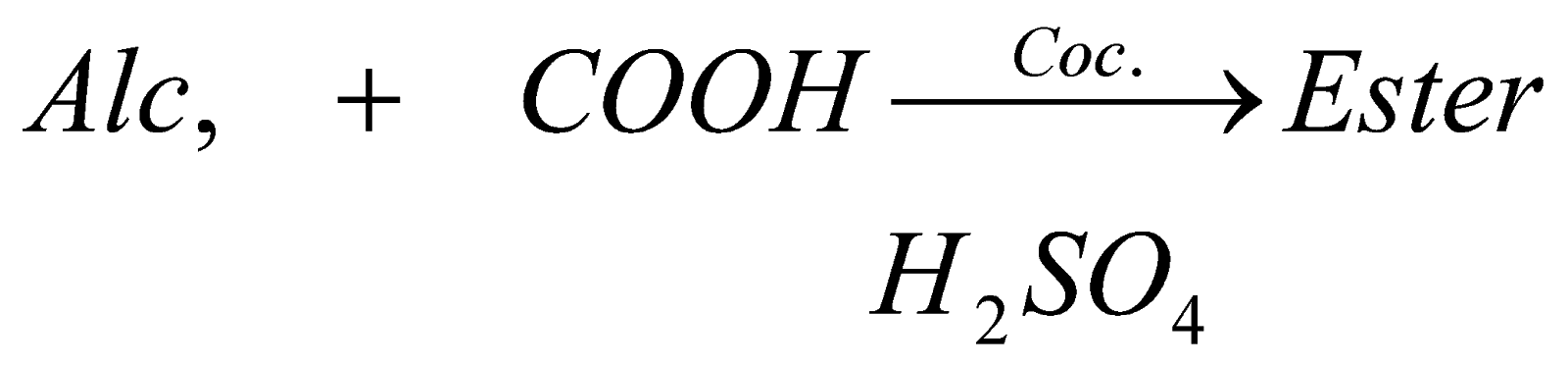
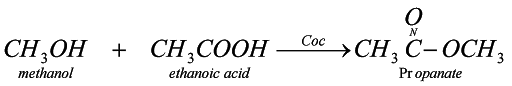
Properties-
Hydrolysis of ester: this is the property when
![]()
Some important reactions-
1) Pyrolysis of alkanes: when alkanes are heated at high temperature at about 1500 degree celcius, they undergo cracking and form lower alkane and an alkene.
It means heating alkanes in absence of air
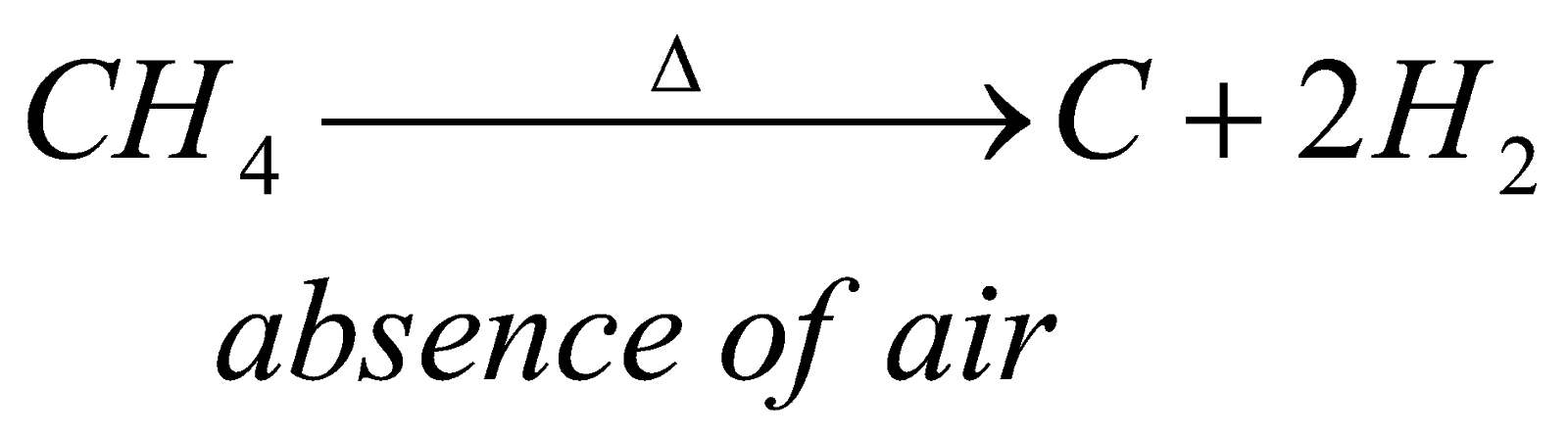 ?
?
C2H6 ? 2C + 3H2
? C6H14——-C4H10 + C2H4
2). Oxidation of alkanes: when alkanes are oxidized in presence of copper they form alcohol and when oxidized in presence of moly carboxylic acid, bdenum oxide, it forms aldehyde and in presence of silver oxide it forms ether.
Alkane ![]()
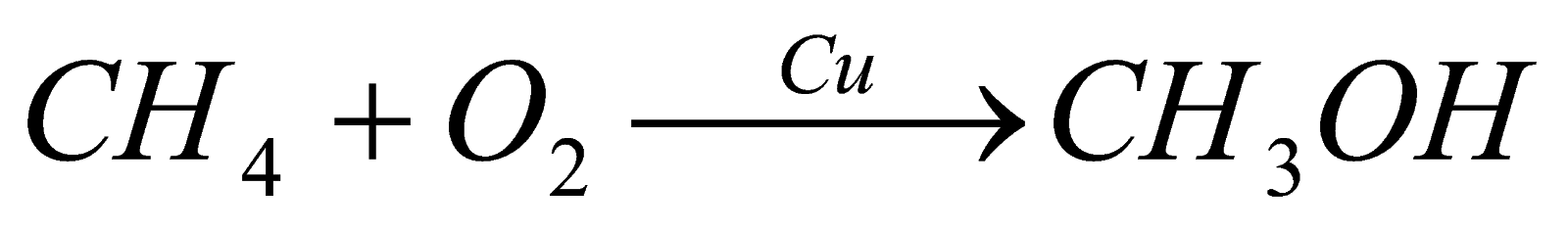
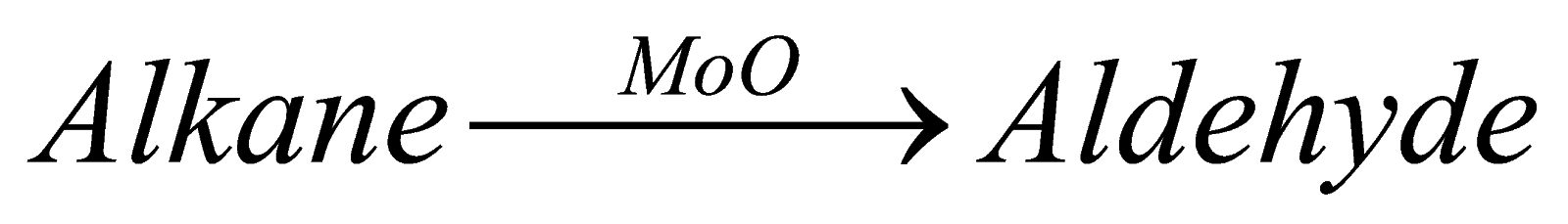
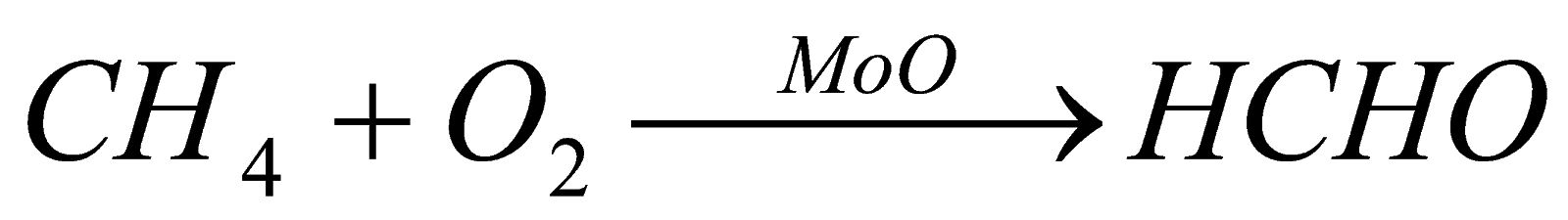
3) Subsitution Reaction : it is the reaction in which hydrogen is substituted by halogen. Like chlorination of methane. This reaction goes on occurying till the time all hydrogs are replaced by chlorine.
CH4 + Cl2 ? CH3Cl + HCl
CH3Cl + Cl2 ? CH2Cl2 + HCl
CH2Cl2 + Cl2 ? CCl3
CHCl3 + Cl2 ? CCl4 + HCl
This is called chlorination of methane and it take place in presence of sunlight.
4) Addition Reaction :
It is shown only by alkenes & alkynes because they have extra bond. So, addition of any substance can be carried out.
i) Addition of Halogen
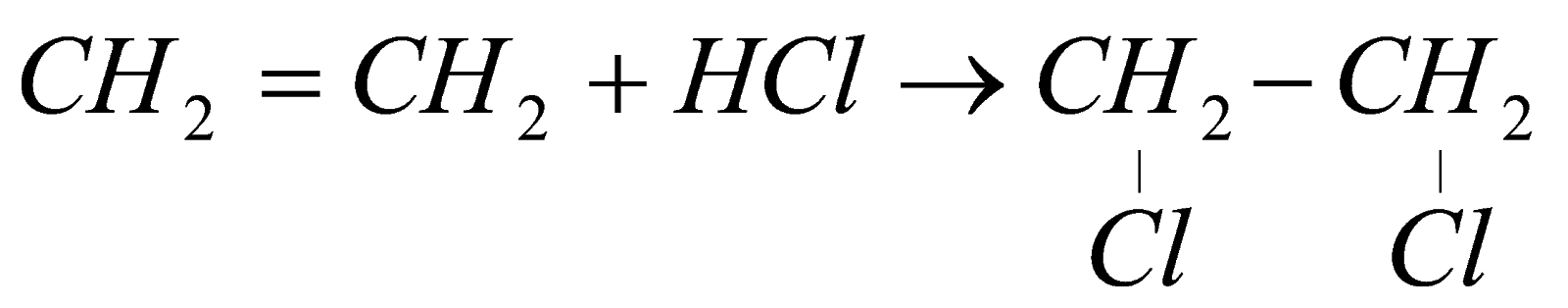
ii) Hydrogenation of oils (Addition of H2): converting liquid vegetable oil into solid fat by adding hydrogen in presence catalyst nickel.
CH2 = CH2 + H2 ![]()
Liquid vegetable oil ![]() fat
fat
5) Combustion- all hydrocarbons, on combustion give, CO2 + H2O + Heat energy + light
Rules for IUPAC
1. Select the longest continuous carbon chain
2. Longest chain with maximum number of subsituents is selected
3. Numbering is done in such a way that substituent gets the lowest number
4. It two substituents are present then we follow the lowest sum rule
5. If the same subsequent appears twice or thrice, then di, tri is used
6. If different substituents are present then alphabetical order is followed
7. If doule/triple bond is present along with substituent then double or triple bond gets lowest number
8. If double and triple bond both represent, lowest number is given to double bond
9. In IUPAC name is written in order
Prefix wordroot.p.suffexsec. suffex
Soaps and detergents

As you all are familiar with soaps, detergents and their cleansing action, so, let us study it in detail.
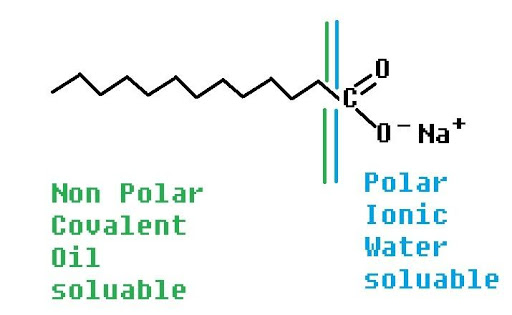
They are sodium salt of long hydrocarbon chain carboxylic acid.
i.e.: RCOONa
Examples
Sodium Stearate
Sodium Palmitate
Preparation
The Raw Materials needed for it are:
- Vegetable oil or animal fat
- Sodium hydroxide
- Pinch of salt
Reaction involved: animal fat or vegetable oil ———-salt? soap + glycerol.
The function of salt is to precipitate soap so that it can be separated from glycerol.
Structure of Soap
Soap has tadpole like structure i.e.:
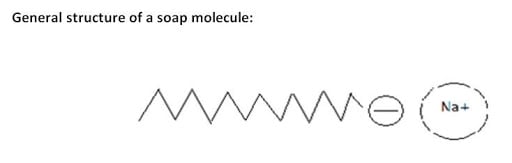
Head is made up of ionic part that is water loving (hydrophilic) and tail is made up of non-ionic part that is water repelling (hydrophobic).
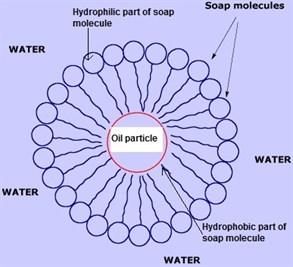
Cleaning action of Soap
In a bucket we take water and add soap to it. Soap molecules gets randomly arranged as shown in figure.
Now when we add dirty cloth to it, soap molecules get arranged in such a way that hydrophobic part moves towards dirt and hydrophilic part moves towards water. This type of arrangement is called micelle formation as shown:
Now due to thermal agitation the different soap molecules attract dirt towards themselves in such a way that it breaks into smaller parts. Hence, dirt remains in water and cloth gets cleaned.
Limitation of using Soap
1.It can’t be used with hard water as hard water contains calcium and magnesium ions. So when soap is used, it interacts with it and forms insoluble precipitate called scum that sticks to cloth. Hence, interferes in cleansing properties of soap.
Advantage of using Soap
It is biodegradable, hence, does not cause any pollution.
Detergents

They are defined as sodium salt of long hydrocarbon chain sulphonic acid.
i.e. RSO3Na
Preparation: It is prepared from petroleum
Structure: Is same like soap i.e. hydrophilic and hydrophobic part

Limitation of using Detergent
It is non-biodegradable, so leads to pollution as its disposal is the main problem.
Difference between soap and detergent
| soap | detergent |
| sodium salt of carboxylic acid | sodium salt of sulphonic acid |
| less soluble in water | more soluble in water |
| less powerful cleansing agent | more powerful cleansing agent |
| required in more amount | required in less amount |
| biodegradable | non biodegradable |
Class 10 Science Chapters, Explanation, Notes
| Life Processes | Chemical Reaction and Equation | Acids bases and Salts |
| Periodic Classification of Elements | Metals and Non-Metals |
NCERT Book Solutions Carbon and its Compounds
Q1. Ethane, with the molecular formula C2H6 has
(a) 6 covalent bonds.
(b) 7 covalent bonds.
(c) 8 covalent bonds.
(d) 9 covalent bonds.
A: (c) 8 covalent bonds.
Q2. Butanone is a four-carbon compound with the functional group
(a) carboxylic acid.
(b) aldehyde.
(c) ketone.
(d) alcohol.
A :(c) ketone.
Q3. While cooking, if the bottom of the vessel is getting blackened on the outside, it means that
(a) the food is not cooked completely.
(b) the fuel is not burning completely.
(c) the fuel is wet.
(d) the fuel is burning completely.
A : (b) the fuel is not burning completely.
Important Videos Links
Q4. Explain the nature of the covalent bond using the bond formation in CH3Cl.
A: The nature of covalent bond : it is formed by mutual sharing of electrons
Q5. Draw the electron dot structures for
(a) ethanoic acid.
(b) H2 S.
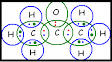
(c) propanone.
(d) F2 .
A. The electron dot structure of :
Ethanoic acid
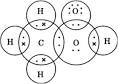
H2S
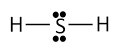
Propanone:
F2
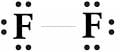
Related – MCQs for Carbon and its Compound Class 10
Q6. What is an homologous series? Explain with an example.
A : Homologous series are the members of same family having same function group,similar properties but they differ their consecutive members by CH2 group and atomic mass by 14 amu.
Examples : homologous series of alkane is :
|
1. |
Methane |
|
2. |
Ethane |
|
3. |
propane |
|
4. |
Butane |
Q7. How can ethanol and ethanoic acid be differentiated on the basis of their physical and chemical properties?
A:
ethanol |
Ethanoic acid |
|
F.gp is -OH |
F.gp is COOH |
|
Ethanol forms sodium ethoxide when treated with sodium |
Ethanoic acid forms sodium ethanoate when treated with sodium |
|
Ethanol does not produce carbon dioxide on treatment with sodium carbonate |
Ethanol produce carbon dioxide on treatment with sodium carbonate . |
Q8. Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents such as ethanol also?
A. Soap has tadpole like structure i.e.:
Head is made up of ionic part that is water loving (hydrophilic) and tail is made up of non-ionic part that is water repelling (hydrophobic).
In bucket we take water and add soap to it. Soap molecules gets randomly arranged as shown in figure:
Now when we add dirty cloth to it, soap molecules get arranged in such a way that hydrophobic part moves towards dirt and hydrophilic part moves towards water. This type of arrangement is called micelle formation.
No, it will not be in ethanol as soaps are soluble in ethanol
Q9. Why are carbon and its compounds used as fuels for most applications?
A :Carbon and its compounds are used as fuels for most applications because when they get oxidised they produce a large amount of energy.
Q10. Explain the formation of scum when hard water is treated with soap.
A: Hard water contains calcium and magnesium ions so when soap is used, it interacts with it and forms an insoluble precipitate called scum that sticks to clothes. Hence, it interferes in cleansing properties.
Q11. What change will you observe if you test soap with litmus paper (red and blue)?
A: When soap is treated with litmus solution it turns it blue .
Q12. What is hydrogenation? What is its industrial application?
A: Hydrogenation is the process in which unsaturated fat is converted in to saturated fat by adding hydrogen in presence of nickel .
It converts liquid vegetable oil to vanaspati ghee .
Q13. Which of the following hydrocarbons undergo addition reactions: C2H6 , C3H8 , C3H6 , C2H2 and CH4 .
A :C3H6,C2H2 wiil undergo addition reactions because they have double bonds.
Q14. Give a test that can be used to differentiate between saturated and unsaturated hydrocarbons.
A: Saturated hydrocarbons when treated with bromine water have no effect but when they are treated with unsaturated hydrocarbons they decolourize bromine water
Q15. Explain the mechanism of the cleaning action of soaps.
A: Soap has tadpole like structure i.e.:
Head is made up of ionic part that is water loving (hydrophilic) and tail is made up of non-ionic part that is water repelling (hydrophobic).In bucket we take water and add soap to it. Soap molecules gets randomly arranged as shown in figure:
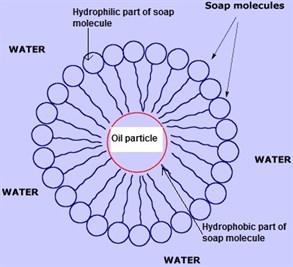
Now when we add dirty cloth to it, soap molecules get arrange in such a way that hydrophobic part towards dirt and hydrophilic towards water this type of arrangement is called micelle formation .
Now due to thermal agitation the different soap molecules attract dirt towards themselves such that it breaks into smaller parts hence remain in water and cloth gets cleaned.
Related – Take Free Online MCQs Science Test
Extra Qustions And Answers
1.Name the product formed other than soap during sponification?
A. Alcohol (glycerol) is the other product formed during sponification.
2.What is the structural formula of ester?
A. 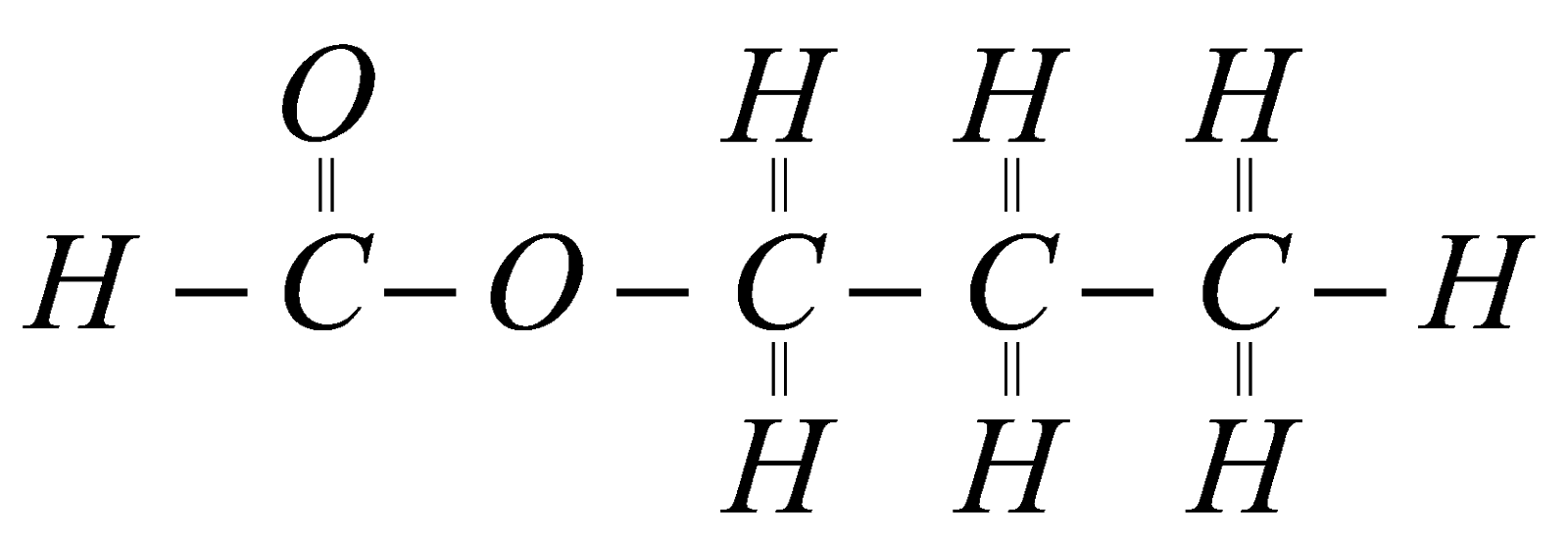
3. Write molecular formula for (a) ethanol (b) propanoic acid ?
A. Ethanol C2H5-OH
propanoic acid CH3CH2COOH
4. What is common name for CH3CHO, HCHO, CH3COOH & CH3COCH3 ?
A. HCHO (formaldehyde) CH3COOH (acetic acid)
CH3CHO (acetaldehyde) CH3COCH3 (acetone)
5. Which two belong to same homologous series?
C2H6O2 , C2H6O , C2H6 , CH4 O
A. C2H6O2 C2H6 C2H6
C2H6O C2H5OH CH4O CH3OH
6.What is the end product of fermentation carried out at 20oC to 30oC ?
A. ethanol
7. Write molecular formula of 3rd member of carbon comp. with gp of CnH2n+1 OH ?
A.Cn H2n+1 OH Alcohols, 3rd mens C3H3x2H-OH => C3H7OH (propanol)
8. Write the name of alcohol derived from pentane?
A. Pentanol
9.How methanol is converted into methanal?
A. Alcohol 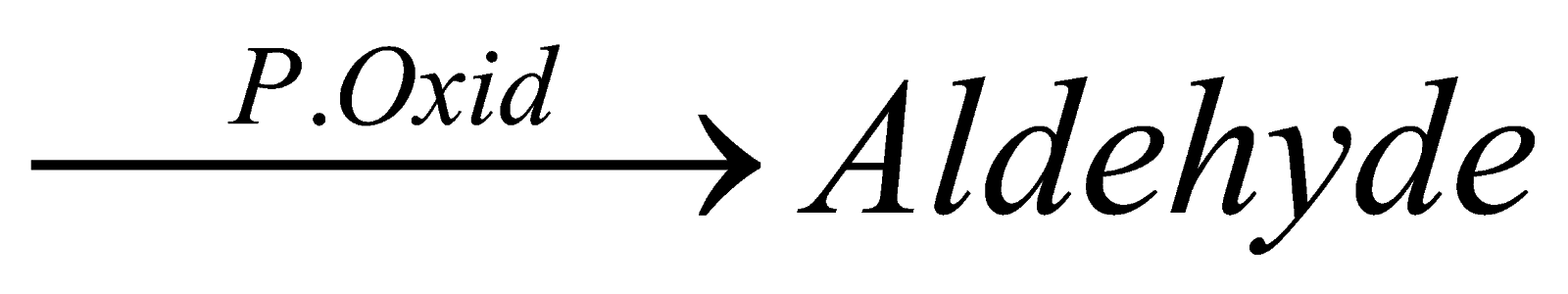 ( By partial oxidation in presence of catalyst)
( By partial oxidation in presence of catalyst)
So, CH3OH 
10. Complete : – C2H5COONa + NaOH ![]()
A. This is decorboxyletion C2H5COONa + NaOH ? 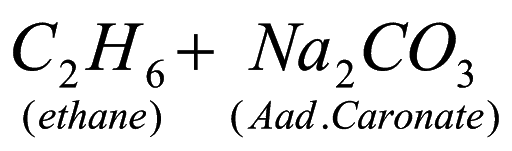
11.Complete : – CH3COOH + C2H5OH 
A. There is esterification . CH3COOH + C2H5OH 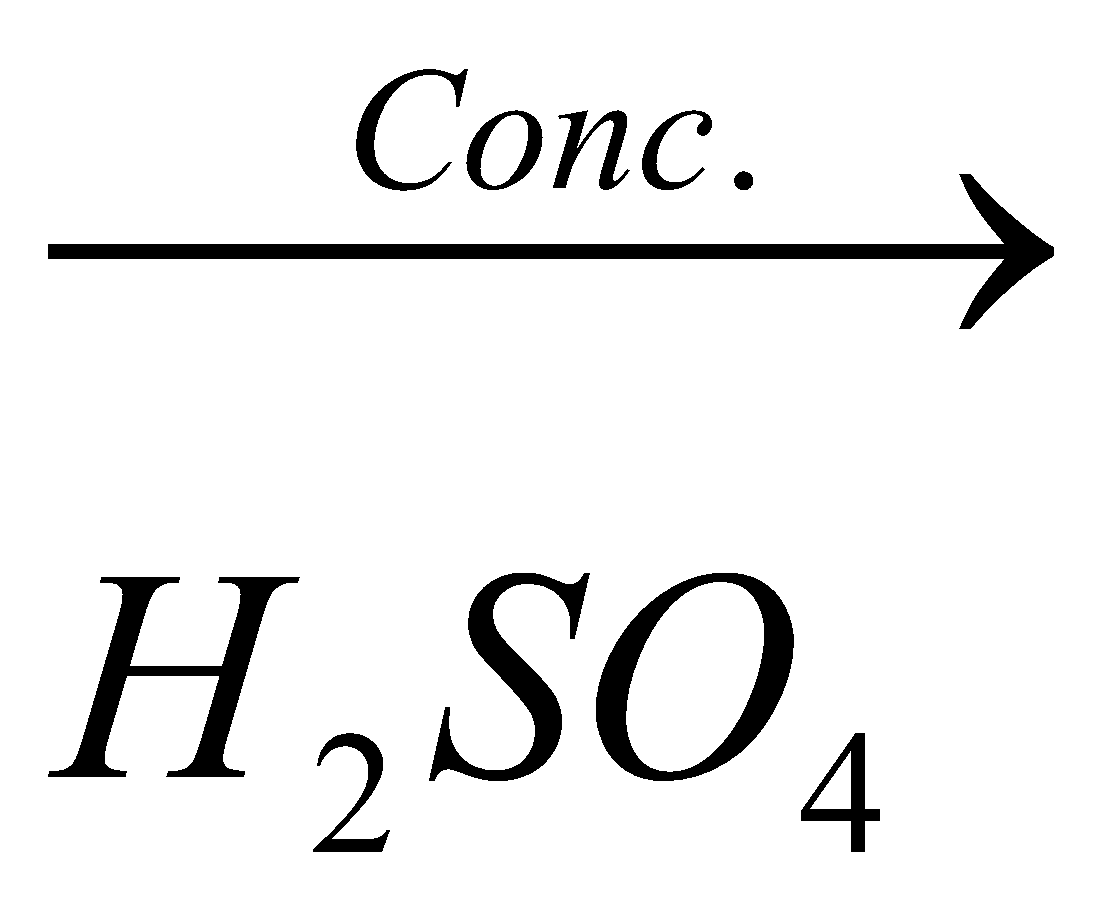
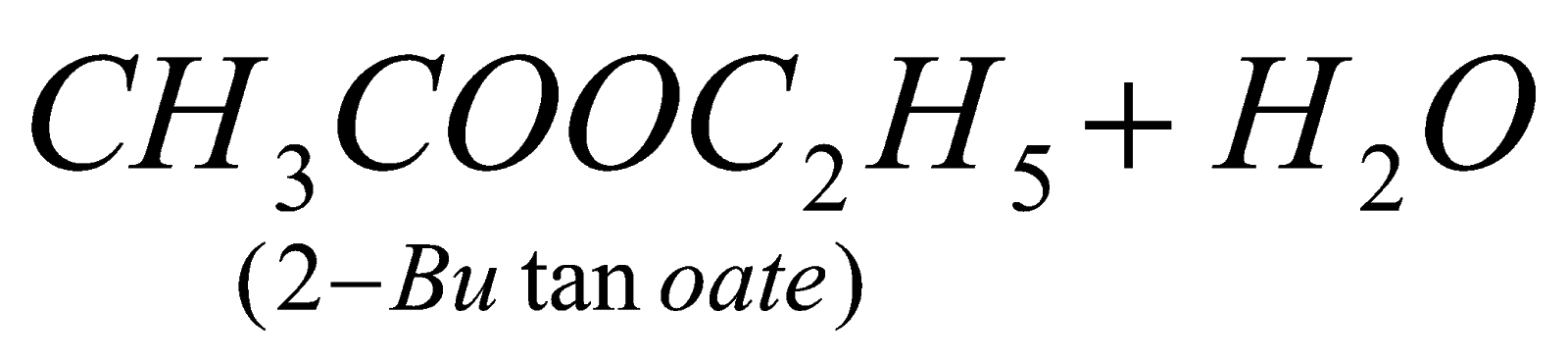
12.What happens when methanol reacts with hydrogen cyanide ?
A. HCHO +HCN ?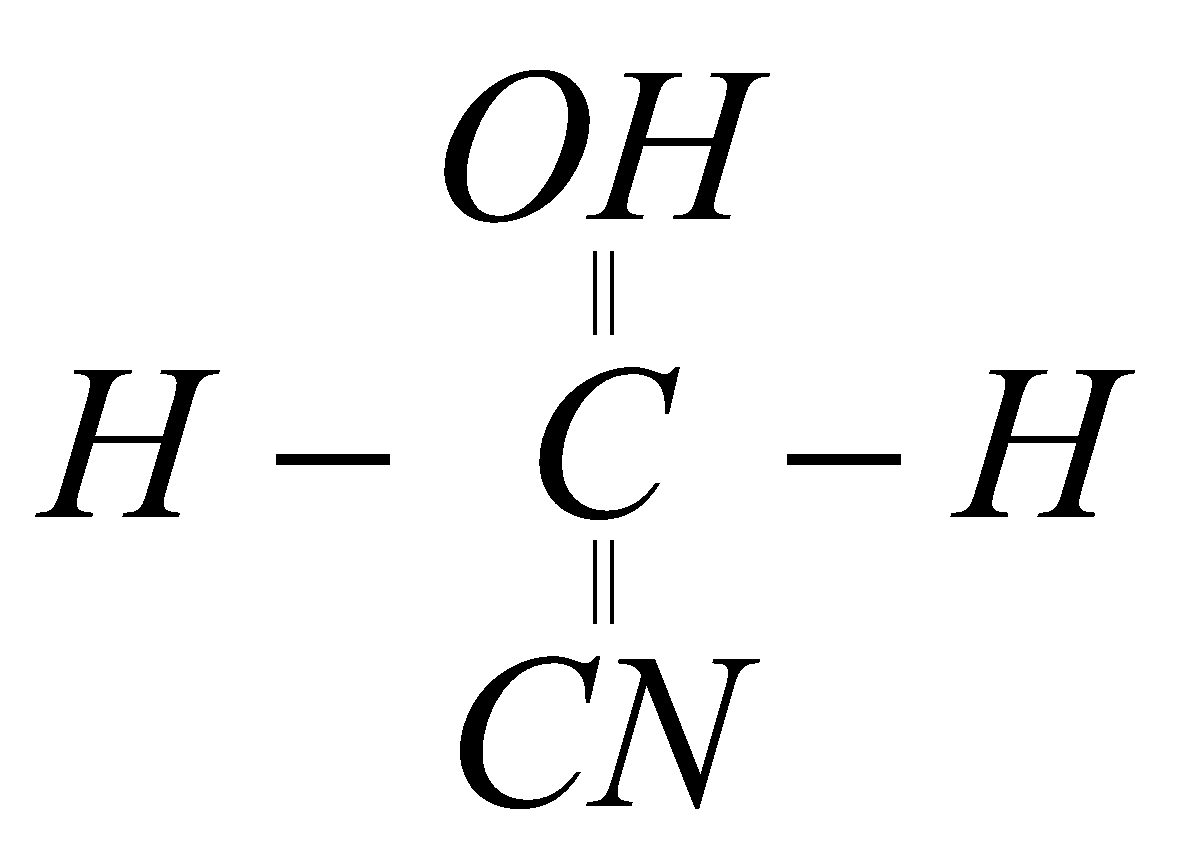
13.What happens when methanol burns with excess of oxygen?
A. 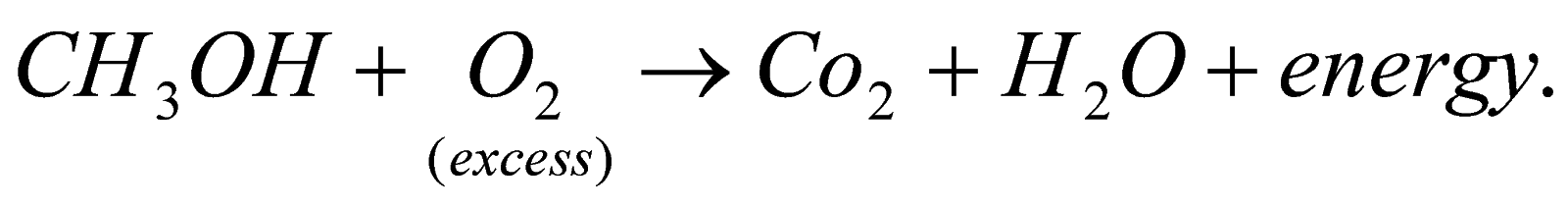
14.Write first member of ketone?
A. CH3COCH3 ( acetone)
15. Name the enzyme that converts glucose to ethanol?
A. Invertase released by yeast.
16. Define sponification.
A. It is the process of formation of soap when animal fat + NaOH –NaCl?soap + glycerol
17. How one can detect diabetes in patient?
A. Diabetic patients excrete sugar crystals in urine. The sugar present is aldehydic sugar. A sample of urine is taken & bendict’s solution is added. If red precipitate is formed that means sugar crystals are present in urine.
18. Convert ethanoic acid to methane?
A. CH3COOH + Na ? CH3COONa + H2
CH3COONa + NaOH ? CH4 + Na2CO3.
19.What is vinegar?
A. Vinegar is acetic acid (ethanoic acid)
20.Give chemical reaction of acetic acid with Na metal?
A. CH3COOH + Na ? CH3COONa + H2
( sodium Ethanoate)
21.Which ions ex present in hard water ?
A. Ca++ & Mg++ ions
22.Why does soap form white ppt. with hard water?
A. Hard water contains Ca++ & Mg++ ions & soap reacts with it to form scum ( white insoluble ppt.)
23. Name the category of compounds formed when one hydrogen of alkane is replaced by hydroxyl gp?
A. Hydroxyl gp;- means OH so, comp,. formed is alcohol
24.A organic compound is formed by reacting unsat.urated hydrocarbon with water in presence of H3PO4. The compound burns with blue flame. Identify compound?
A. Lens. Hydrocarbon + H2O 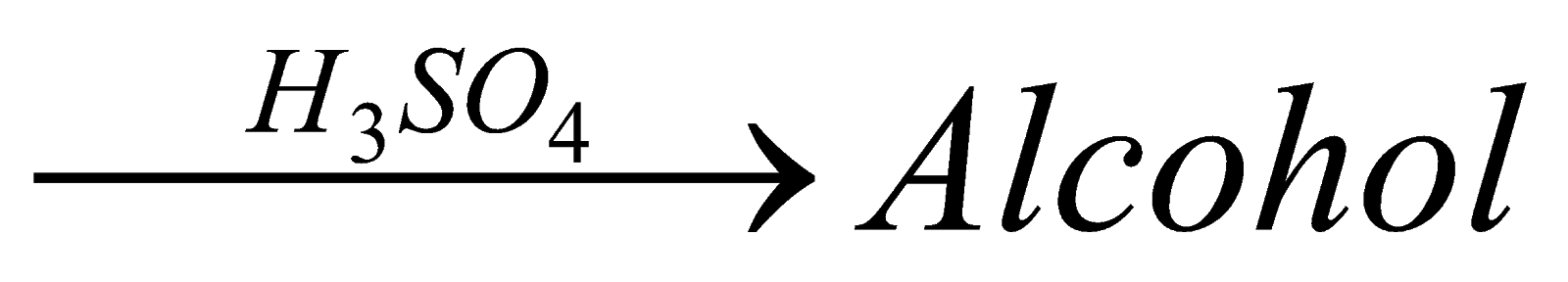
Eg. Hydration of ethene
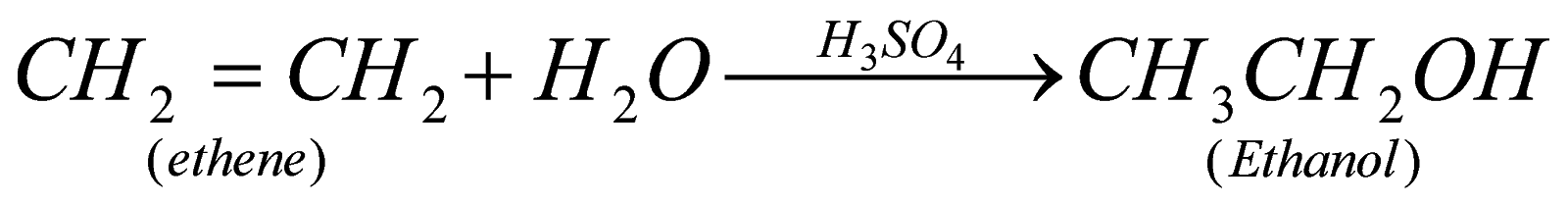
25. What is formalin? Name product formed when it is treated with fehling’s solution?
A. Formalin is 35-40 % solution of formaldehyde (HCHO) in water. When it is treated with fehling’s, it forms- formic acid (HCOOH) as it gets oxidized.
26.Give the names : –
A. (a)Aldehyde formed from ethane ? ethanal
(b) comp. formed by oxidation of ethanol with CrO3 ? ethanal
27.Write reaction of sodium with ethanol ?
A. C2H5OH + Na ? C2H5ONa + H2
(sod. Ethoxide)
28. Convert acetic acid to methane ?
A.CH3COOH + Na ? CH3COONa + H2
CH3COONa + NaOH ? CH4 + Na2CO3
29. Define hydrogenation of oils?
A.The process of heating liquid veg. oils with H2 is process of hydrogenation which gives vanaspati ghee.
30. Complete CH2 = CH2 + H2 ![]() ?
?
A. 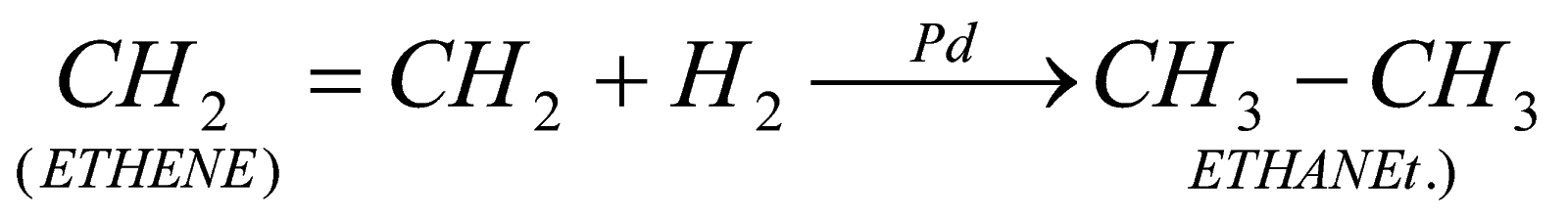
31.Out of CH3OH, C2OH, C2H5OH which her lowest melting point?
A. A compound with less number of carbon atoms has lowest m.pt. i.e CH3OH .
32.What determines the property of organic compound?
A. F.gp.
33.How can you show CH3COOH is acidic in nature?
A. Add litmus solution, if it turns red it shows it is acidic in nature
34. Which of them will undergo addition rxn C2H6, C3H8, C3H6, C2H2, CH4?
A. Only unsaturated hydrocarbon will undergoes addition rxn i.e. C3H6, C2H2