CBSE Class 10 Science Notes of Chapter 3 Metals and Non-Metals, Video Explanation and Question Answers
Metals and Non-Metals Class 10 Notes – Chemical Reactions and Equations Class 10 Science Chapter 3 as per NCERT Book used in CBSE and other Schools. The lesson covers the complete explanation of Class 10 Science Chapter 3 Metals and Non-Metals.
Here is a CBSE Class 10 Science Chapter 3 Metals and Non-Metals Notes and NCERT Solutions to Important Question Answers. Given here is the complete explanation of the chapter, along with examples and all the exercises, Questions and Answers are given at the back of the chapter.
Topics covered are Metals and Non-Metals Video Explanation, Metals, Metallurgy, Non metals, Chemical Bonding, Physical Properties of metals and non metals, Alloys, Chemical Properties of metals and non metals and NCERT Solutions Class 10 Science Chapter 3.
The lesson covers all important questions and NCERT solutions
Class 10 Science
Chapter 3 – Metals and Non-Metals

- See Video Explanation of Metals and Non-Metals
- metals
- metallurgy
- non metals
- chemical bonding
- physical properties of metals and non metals
- alloys
- chemical properties of metals and non metals
- Metals and Non-Metals Class 10 NCERT Solutions
Element: Is a substance that can’t be broken into more simpler substances. Elements are classified into different categories:
- Metals
- Non Metals
- Metalloids
- Noble Gases
Metals
Metals are those elements which have 1,2 or 3 valence electrons in their valence shell.
Example : atomic no. of Na =11
Electronic confugration =2,8,1
They always loose electrons to form electropositive ions.
Na – 1electron ——Na +
Non Metals
Non metals are those elements which have 4,5,6 or 7 valence electrons in their valence shell and they always gain electrons to form electronegative ions.
Cl(17) – 1 electron ——–Cl-
Electronic Configuration = 2,8,7
Related – Tips to score 95% in Class 10 Science Paper
Metals and Non-Metals Video Explanation
Physical Properties Of Metals and Non Metals
1. malleability: process of beating into thin sheets
Metals :are malleable
Non metals :non malleable because they are brittle
2. ductility: process of drawing into thin wires
Metals:ductile
Non metals: nonductile because they are brittle
3. Electrical Conductivity And Thermal Conductivity:
Metals: good conductor of heat and electricity because of free mobile electrons in it.
Non metals: poor conductor of heat and electricity.
4. lustre: shine
Metals: lustrous
Non metals: non lustrous
5. Hardness:
Metals: hard
Non metals: soft
6. physical state:
Metals: solid at room temperature
Non metal: can be solid, liquid or gaseous
7. melting and boiling point:
Metals :high m.pt and b.pt
Non metals: low m.pt and b.pt
8. Sonority: when hit, they produce sound
Metals:sonorous
Non metal:non sonorous
Chemical properties of metals and non metals

1 Reaction With Air
Metals react with oxygen to form metal oxide
metal +oxygen → metal oxide
Na + O2→ Na2O
Mg+ O2→ MgO
If we notice the nature of oxides the Metal oxides are basic in nature, i.e., they turn red litmus blue but some metal oxides proved to be amphoteric (i.e they have acidic and basic character). For example aluminium oxide and zinc oxide.
We can show their amphoteric nature by reacting them with acid and base i.e.

AL2O3+NaOH→ NaAlO2 +H2O
AL2O3+ HCl → AlCl3+ H2O
Order of reactivity with oxygen:Na>Mg>Zn>Fe>Cu
- Sodium and potassium are stored in kerosene oil because when they come in contact with oxygen, they form respective oxides and the reaction is explosive. So, in order to avoid contact with air, it is stored in kerosene oil.
Non metals react with oxygen to form non metal oxide
Non Metal +Oxygen →Non Metal Oxide
C+ O2→CO2
N2+O2→NO2
If we notice about Nature of non metal oxides, they are acidic i.e., turn blue litmus red. But Some non metal oxides are neutral like water, carbon monoxide, etc
Reactivity Series – Arrangement of elements according to their reactivity
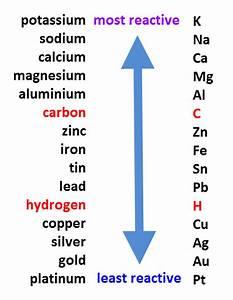

2 Reaction with water
Metal react with water forming metal hydroxide and hydrogen gas is released
i.e., metal +water →metal hydroxide +hydrogen gas
Na + H2O→NaOH+H2
Some react with cold water: sodium and potassium

Some react with hot water: calcium and magnesium
Some react with steam: iron, zinc and aluminium
when iron reacts with steam : Fe + H2o→Fe3O4+H2
Some donot react: elements below hydrogen in reactivity series like copper, mercury, silver, gold and platinum. Look at the picture copper in water remain as it :
Metals below hydrogen are less reactive than hydrogen so can’t displace hydrogen from acid.
Order of reactivity:Na>Mg>Zn>Fe>Cu
Reaction of non metals with water: Non metals do not react with water as to react, they need to displace hydrogen and for displacing, they need to donate electrons to hydrogen but it is not possible as they are themselves electron acceptors.
3 Reaction with dilute acids
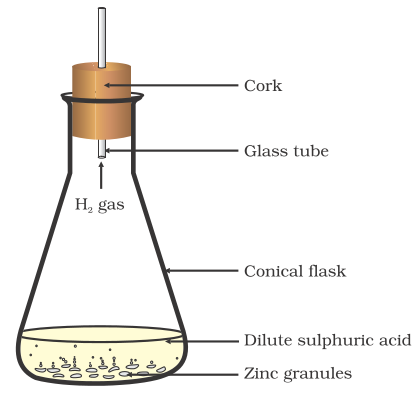
Reaction of metals with dilute acids
Metals react with acids to form respective salt and hydrogen gas is released
for example H2So4+Zn→ ZnSo4+H2
Metals below hydrogen in the reactivity series can’t displace hydrogen of acids as they are less reactive than hydrogen.
Non metals do not react as they can’t displace hydrogen from acid. For displacing, they need to donate electrons to hydrogen but it is not possible as they are themselves electron acceptors.
Class 10 Science Chapters
METALLURGY
As we know, metals have good reactivity. So, they generally occur in combined state but few metals that are less reactive occur in free state like platinum ,gold etc. So, let us study about occurrence of metals and how they are extracted.
Minerals: Anything that is found in earth’s crust is called mineral.
Ores: The mineral from which metal can be extracted is called ore.
Note: All ores are minerals but all minerals are not ores.
Different Types of Ores:
-
- Oxide ores
- Halide ores
- Sulphide ores
- Carbonate ores
Ores of Some Metals:
- Aluminium: Bauxite ore, cryolite ore
- Iron: Haemtite ore, magnetite ore
- Copper: Copper pyrite, copper glance
- Zinc: Zinc Blende
- Mercury: Cinnabar
Metallurgy is the process of extracting metal from its ore.
Various steps involved in metallurgy are as follows –
- Concentration of ore
- Oxidation of ore
- Reduction of ore
- Refining
Let us discuss them in detail:
Step 1
The Concentration of ore: It involves various methods like hydraulic washing, magnetic separation, froth flotation, chemical separation
Hydraulic Washing: In this, the high-speed stream of water is passed through a slope which has ore placed on it. As a result, the heavier ore stays behind and lighter impurities flow along with the stream of water.
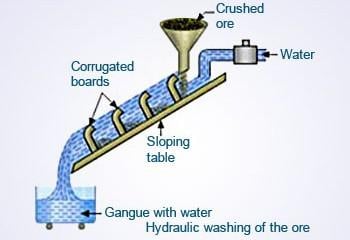
Limitation of this method: It is used only for heavy ores like iron
Magnetic Separation: In this apparatus is set as shown in figure.
The powdered ore containing impurities is poured on a slope with the help of hopper. As the leather belt revolves, it also revolves along. As a result, ore falls later as it is being attracted by magnet and impurities falls first and they form different heaps. Hence, they get separated.
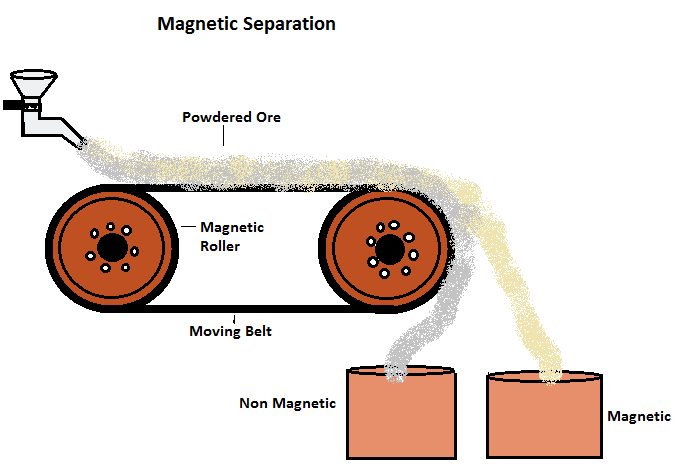
Limitation: It is used for ores only with magnetic properties
Froth Floatation: In this the apparatus is set as shown in figure:
The powdered ore containing impurities is poured in the tank containg pine oil and water. Now with the help of compressor, compressed air is passed. As a result, froth appears to which ore sticks and impurities are left behind.
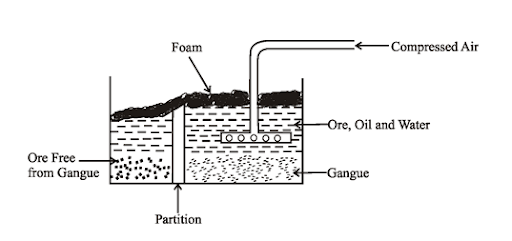
Limitation: Only used for sulphide ores as they have attraction towards pine oil froath
Chemical Separation: It is the method in which some chemical substance is added to the powdered ore containing impurities. As a result, that chemical substance only reacts with ore but not with impurities. For example Bayer’s process in case of aluminium ore
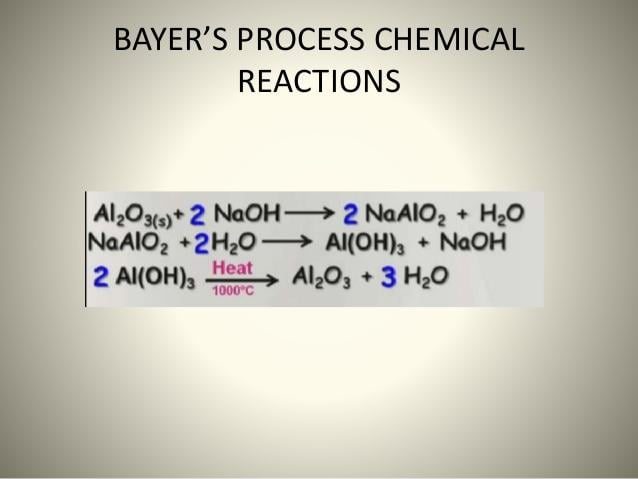
Step 2
Oxidation of ore: It means converting concentrated ore into metal oxide.
The methods involved are calcination and roasting
- Calcination
- Roasting
Calcination: is the process of heating ore in absence of air
Ore →metal oxide
Example : ZnCo3→ZnO +Co2
This method is used only for carbonate ores
Roasting: it is the process of Heating ore in presence of air
Ore →air→metal oxide
This is used for sulphide ores
For Example:
ZnS—–(heated in air )→ZnO+So2
Step 3
Reduction of metal
It means converting metal oxide into metal by using various processes
The methods that can be employed are:
- By heating
- By using reducing agent
- By using electric current: Electrolytic reduction
By Heating: This method is used for those metals that lie at the bottom of the reactivity series
Metal Oxide ———- heat →metal + oxygen
For Example: HgO→Hg +O2
By use of reducing Agent: Mostly C or Al is used as reducing agent
This method is used for those metals that lie in mid of reactivity series
In this metal oxide → C or Al→ metal + oxygen
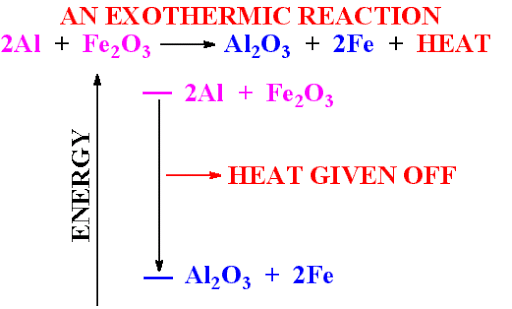
For Example: this reaction is commonly used in welding of railway tracks
By Electrolysis: This method is used for elements that occur on the top of reactivity series first we should know about electrolysis
The process in which an electrolyte dissociates into ions when the current is passed through it is called electrolysis.
 /p>
/p>
Electrode:
- Cathode: negative terminal
- Anode: positive terminal
- Electrolyte: A substance that dissociate into ions
The Chemical reactions take place and as a result various observations can be seen:
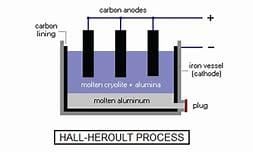
Let us explain by taking an example of Electrolysis of aluminium oxide:
 /p>
/p>
- Cathode: Carbon electrode
- Anode: carbon electrode
- Electrolyte: Aluminium oxide and to it cryolite and feldspar is added as they lower the melting point of aluminium oxide and also increase its degree of dissociation.
On passing Current, we get-
Aluminium oxide→AL3+ +O2-
The ions move to respective poles and following reaction occurs-
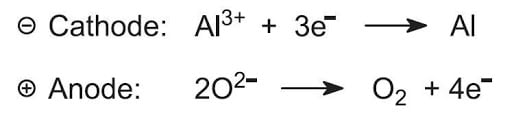
The impurity that collects under anode is anode mud.
Step 4
Refining of metal
The various methods used are:
Liquation, electrolytic refining
Liquation method: The apparatus is set as shown in figure
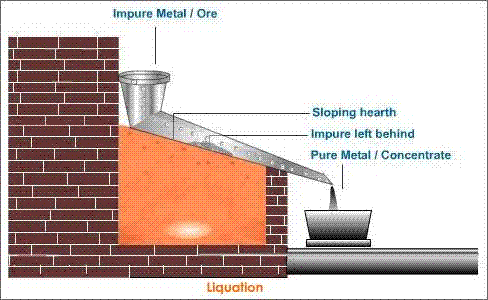
In this, the metal is placed on a hearth and is heated from below. As a result, metal melts and flows down due to its low melting point in comparison to impurities and impurities are left behind. Hence, the metal obtained is refined metal.
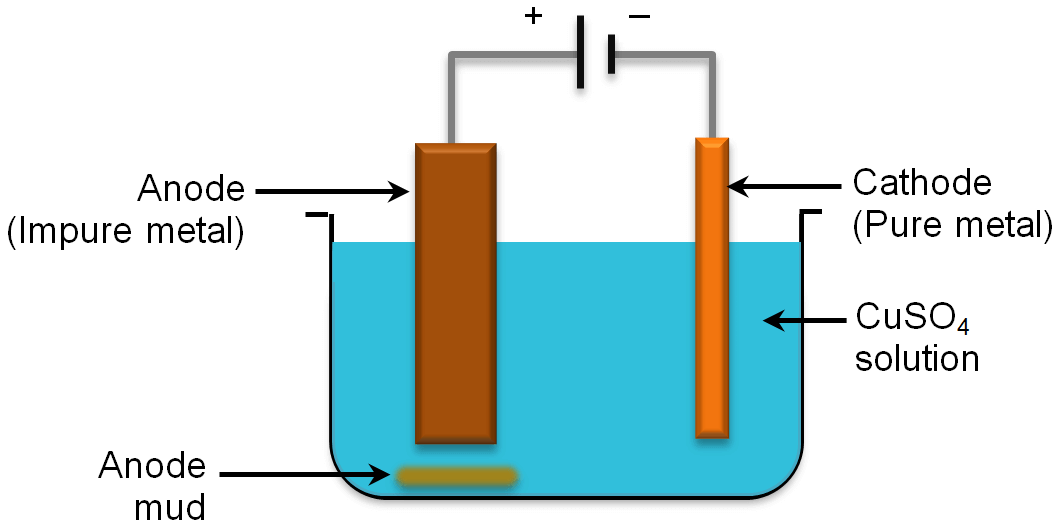
Electrolytic Refining
Cathode: Pure metal (Cu)
Anode: Impure metal (Cu)
Electrolyte: salt of metal to be refined (Cuprous oxide and Cu2S)
Mechanism: On passing current the electrolyte dissociates into ions i.e. These ions migrate to respective poles
Reaction at cathode
Reaction at anode
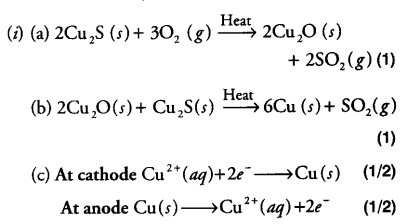
Let us explain by taking an example of copper
CHEMICAL BONDING
It is linkage between two or more atoms in a molecule.
Cause: The atoms combine with one another to achieve noble gas configuration. By doing so they become stable.
Atom can achieve noble gas confugration
- By loosing one or more e’s.
- By gaining one or more e’s.
- By sharing one or more e’s.
Types of bonds-
- IONIC BOND (or electrovalent)
- COVALENT BOND
IONIC BOND
- Bond formed by transfer of electrons from one atoms to another.
- It is formed when one atom donate electron & other accept electrons to achieve stable configuration.
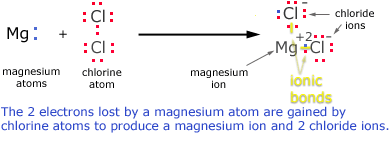
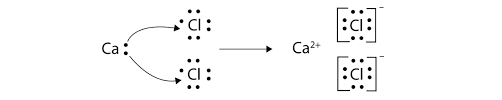
- NOTE: It is always formed between a metal and a non-metal

COVALENT BOND
- It is formed by sharing of electrons between atoms
- It is always formed between non-metal & non-metal.
Types of Covalent Bonds
- Single Covalent bond
- Double Covalent bond
- Triple Covalent bond
Single Covalent Bond: Formed by sharing of one pair of electrons between two atoms
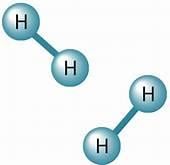
Double Bond: Bond is formed by sharing of two pairs of electron between two atoms.
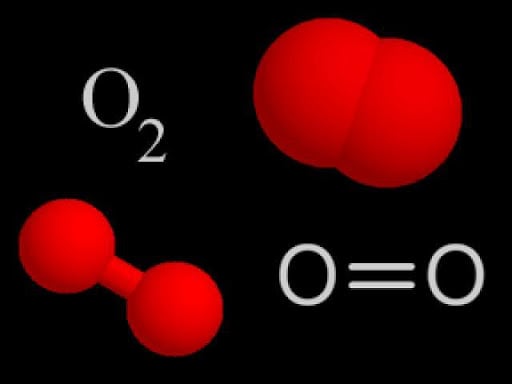
Triple Bond: Bond formed by sharing of three pairs of electrons between two atoms.
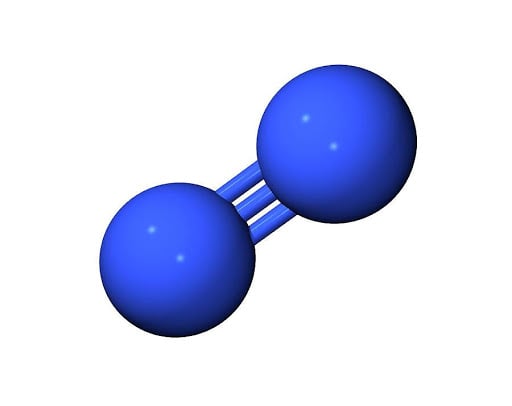
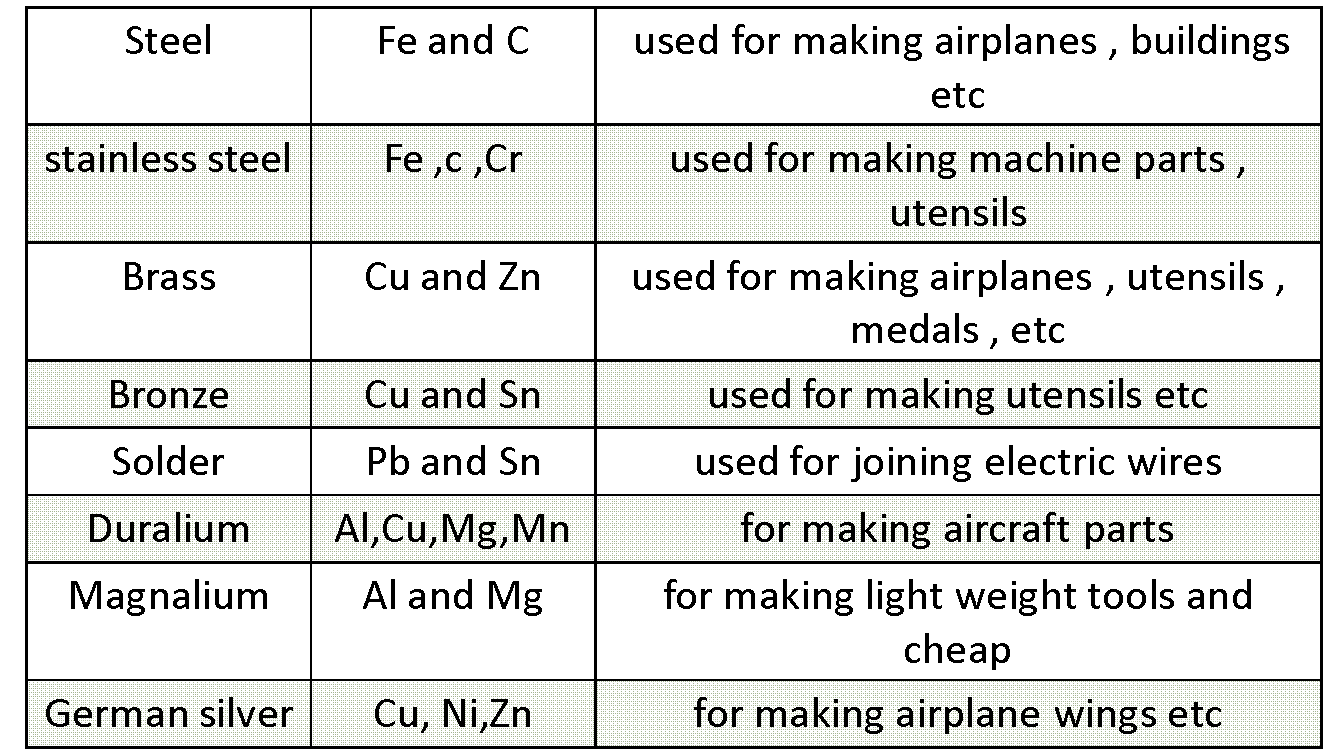
Alloys
A homogenous mixture of two or more metals is an alloy.
- They have properties better than an Metal
Let us see few alloys with their properties and uses:
Class 10 Science Chapters, Explanation, Notes
| Life Processes | Chemical Reaction and Equation | Carbon and it’s compound |
| Periodic Classification of Elements | Acids Bases and Salts |
NCERT Solutions Class 10 Metals and Non-Metals
Q1. Which of the following pairs will give displacement reactions?
(a) NaCl solution and copper metal
(b) MgCl2 solution and aluminium metal
(c) FeSO4 solution and silver metal
(d) AgNO3 solution and copper metal.
A : (d) AgNO3 solution and copper metal.
Q2. Which of the following methods is suitable for preventing an iron frying pan from rusting? (a) Applying grease
(b) Applying paint
(c) Applying a coating of zinc
(d) All of the above.
A : (d) All of the above.
Q3. An element reacts with oxygen to give a compound with a high melting point. This compound is also soluble in water. The element is likely to be
(a) calcium
(b) carbon
(c) silicon
(d) iron.
A :(a) calcium
Important Videos Links
Q4. Food cans are coated with tin and not with zinc because
(a) zinc is costlier than tin.
(b) zinc has a higher melting point than tin.
(c) zinc is more reactive than tin.
(d) zinc is less reactive than tin.
A : (c) zinc is more reactive than tin.
Q5. You are given a hammer, a battery, a bulb, wires and a switch.
(a) How could you use them to distinguish between samples of metals and non-metals?
(b) Assess the usefulness of these tests in distinguishing between metals and non-metals.
A. Metals : on hammering it will not break
When circuit is made while connecting metal, bulb and switch; the bulb glows as it is a good conductor of electricity.
Non metals : on hammering it breaks
On connecting it to circuit, bulb doesn’t glow.
Related – MCQs Chapter 3 Metals and Non-Metals Class 10
Q6. What are amphoteric oxides? Give two examples of amphoteric oxides.
A :some metal oxides have acidic and basic character. For example aluminium oxide and zinc oxide .
We can show their amphoteric nature by reacting them with acid and base i.e
AL2O3+NaOH→NaAlO2 +H2O
AL2O3+ HCl→AlCl3+ H2O
Q7. Name two metals which will displace hydrogen from dilute acids, and two metals which
will not.
A : metals : sodium and calcium
Metals that do not react : copper and silver
Q8. In the electrolytic refining of a metal M, what would you take as the anode, the cathode and the electrolyte?
Ans :anode : impure metal
Cathode : pure metal
Electrolyte : metal salt solution
Q9. Pratyush took sulphur powder on a spatula and heated it. He collected the gas evolved by inverting a test tube over it, as shown in figure below.
(a) What will be the action of gas on
(i) dry litmus paper?
(ii) moist litmus paper?
(b) Write a balanced chemical equation for the reaction taking place.
A : (i) no effect on dry litmus
(ii) Moist litmus : turns red litmus blue
(b) S+O2–>SO2
Q10. State two ways to prevent the rusting of iron.
Ans : 1.galvanisation
2. Alloying
Q11. What type of oxides are formed when non-metals combine with oxygen?
Ans : Acidic oxides and few form neutral oxides
Q12. Give reasons
(a) Platinum, gold and silver are used to make jewellery.
(b) Sodium, potassium and lithium are stored under oil.
(c) Aluminium is a highly reactive metal, yet it is used to make utensils for cooking.
(d) Carbonate and sulphide ores are usually converted into oxides during the process of extraction.
A : (a)Platinum, gold and silver are used to make jewellery because they are inert
(b) Sodium, potassium and lithium are stored under oil because when they come in contact with air they form their oxides which are explosive in nature
(c) it is used in making utensils as it is malleable
(d) yes they are converted in to oxides because it is easy to obtain metal from their oxides
Q13. You must have seen tarnished copper vessels being cleaned with lemon or tamarind juice. Explain why these sour substances are effective in cleaning the vessels.
A : Ontarnished copper there is a layer formed on it which is basic so when we react it with lemon that contain acid the neutralization reaction happens .
Q14. Differentiate between metal and non-metal on the basis of their chemical properties.
|
Metal |
Non metal |
|
1.They readily react with oxygen to form oxides which are basic |
|
|
2. They react readily to form hydroxides |
2. They do not react with water |
|
3. They react with dilute acids forming respective salts and liberate hydrogen gas |
3. They do not react with dilute acids |
|
4. They form ionic hydrides |
4. They form covalent hydrides |
Q15. A man went door to door posing as a goldsmith. He promised to bring back the glitter of old and dull gold ornaments. An unsuspecting lady gave a set of gold bangles to him which he dipped in a particular solution. The bangles sparkled like new but their weight was reduced drastically. The lady was upset but after a futile argument the man beat a hasty retreat. Can you play the detective to find out the nature of the solution he had used?
A : He had used the solution of aqua regia which is actually a mixture of Nitric acid and hydrochloric acid in ratio 3:1 and it can dissolve even noble metals .
Q16. Give reasons why copper is used to make hot water tanks and not steel (an alloy of iron).
A : steel alloy of iron is not used as it is more reactive than copper it reacts with hot water forming ferrosoferric oxide due to which the body of tank will become weaker .
Related – Take Free Online MCQs Science Test
Extra Question and Answers
Q1. Which of the following pairs give displacement reaction ?
- sodium chloride and copper metal
- magnessium chloride and aluminium metal
- ferrous sulphate nad silver metal
- silver nitrate and copper metal ?
A. Out of them, fourth – silver nitrate and copper metal gives displacement reaction as in that case the more reactive metal displaces the less reactive metal from its compounds. The equation is as follows –
AgNO3+Cu —-Cu(NO3)2 + Ag
Q2. Which of following methods is suitable for preventing an iron frying pan from rusting?
A. Applying a coating of zinc will be a better method as its melting point is less than that of iron so it can bear the heat also.
Q3. An element reacts with oxygen to give a compound with high melting point and this compound is also soluble in water. The element is likely to be : calcium , carbon, silicon or iron ?
A. The element is Calcium.
Q4. Why food cans are coated with tin and not with zinc?
A. As zinc is more reactive than tin and can react with biochemicals of food.
Q5. What are amphoteric oxides. Give example?
A. The oxides that possess acidic as well as basic character are amphoteric in nature .
For example : aluminium oxide and zinc oxide
Q6. In electrolytic refining of metal “m”, what would you take as anode , cathode and electrolyte?
A. In electrolytic refining of metal “m”,
anode –thick impure metal plate.
cathode – thin plate of pure metal.
electrolyte : soluble salt of metal “m”.
Q7. Give reasons:
a) Gold and silver are use in making jewellery.
b) Aluminium is reactive but still used for making utensils and cooking.
A.
a) Gold and silver are used in making jewellery as they are ductile , malleable and non rusting
b) Aluminium is strong and cheap metal it is reactiive also but when it comes in contact with oxygen it forms layer of oxide on its surface that do not let any corroding hence it is used to make utensils and for cooking.
Q8. What are alloys?
A. Alloy is a homogenous mixture of two or metals. They have properties better than the metal. For example brass , bronze, etc.
Q: what is thermite reaction? give its one use.
A. A reaction in which iron oxide is reduced by aluminium is a thermite reaction.
that is : Fe2o3+ A l —-Al2o3 + Fe
in this reaction heat is evolved and it is so enormous that it is used for welding purpose.
Q10. Predict the type of bonding in carbondioxide, aluminium chloride and ethyne?
A. In carbondioxide – double covalent bond
In aluminium chloride : ionic
In ethyne : double covalent bond
Important Questions Videos Links