CBSE Class 10 Science Notes of Chapter 2 Acids Bases and Salts Video Explanation and Question Answers
Acids, Bases and Salts Class 10 Notes – Acids, Bases and Salts Notes of CBSE Class 10 Science Chapter with detailed video explanation of the chapter ‘Acids, bases and salts’ along with Concepts explaining. Given here is the complete explanation of the chapter, along with examples and all the exercises, Question and Answers given at the back of the chapter.
Here is a CBSE Class 10 Science Chapter 2 Acids, Bases and Salts Notes and NCERT Solutions to Important Question Answers. Given here is the complete explanation of the chapter, along with examples and all the exercises, Questions and Answers are given at the back of the chapter.
Topics covered are Acids, Washing Soda, Bases, Baking Soda, Physical properties of acids and bases, Plaster of Paris, Chemical Properties of acids and bases, Bleaching Powder, Ph and its biological importance, indicators and NCERT Book Solutions.
The lesson covers all important questions and NCERT solutions to book questions have also been provided for the convenience of the students.
Class 10 Science
Chapter 2 Acids Bases and Salts Notes

- See Video Explanation of Acids Bases and Salts
- Acids
- washing soda
- bases
- baking soda
- physical properties of acids and bases
- Plaster Of Paris
- chemical properties of acids and bases
- Bleaching powder
- Ph and its biological importance, indicators
- NCERT Book solutions
Acids Bases and Salts Video Explanation
Acids, Bases and Salts Chapter Introduction
Like we individuals have certain kind of nature, likewise, every chemical substance that we come across has some nature. But in terms of chemicals, we relate nature either with acidic, basic or neutral nature.
But to know exactly what is acidic, basic or neutral, we need to know about what exactly we mean by being acidic, basic or neutral. So, accordingly, different concepts were introduced to define them. So, let us study them first .
Earlier, it was thought that those substances that are sour, turn blue litmus red and are corrosive are called acids.
Whereas those which are bitter, turn red litmus blue, soapy in touch are called bases.
But later this concept was replaced by relevant concepts like lewis concept, bronsted lowry concept and Arrhenius concept.
In our syllabus we need to know only about Arrhenius concept .

Acids
Arrhenius Concept: According to his way of judging substances, he classified the acids and bases as given below:
Acids are those that when dissolved in water, give hydrogen ion or we can write hydronium ion (H3O+)
This hydronium ion is formed due to the combination of hydrogen ion produced by acid with water molecule.
Related – Tips to score 95% in Class 10 Science Paper
Example of acids are:
- hydrochloric acid (HCl)
- sulphuric acid (H2SO4)
- nitric acid (HNO3)
- phosphoric acid (H3PO4)
- carbonic acid (H2CO3)
Classification Of Acids: we have different classification based on different factors so let’s sum up and make ourselves familiar with it.
1. Depending upon source from which they are obtained
- Organic acids
- Inorganic acids
Organic Acids are obtained from plants and animals or we can say they are present in organic matter.
Example: in tomato, oxalic acid is present , in apple, malic acid and in lemon, we have citric acid and so many other acids are present in different organic substances.

Inorganic Acids are those that are obtained from minerals present in earth. These are quite reactive in nature.
Example we have are : nitric acid, sulphuric acid, etc.

2. Classification of acids on the basis of strength
If we talk about strength, it means the amount of hydrogen ions given out when acid is dissolved in water.
On the basis of this we have two categories of acids –
- Strong acids
- Weak acids
Strong Acid is the acid that completely dissociates into hydrogen ion. These acids
totally dissociate and leave no dissociated molecule of acid.
Example: Hydrochloric acid, sulphuric acid, etc.
Weak Acid is the acid that partially dissociates into hydrogen ions. There actually
exists the equilibrium between dissociated ions and undissociated molecules of
acids .
Example: Carbonic acid.
3. Classification of acids on the basis of water content
- Dilute acid
- Concentrated acid
- Dilute Acid is the acid that has more amount of water in it and less salt content.
- They are not quite strong.
Concentrated Acid is that which has less amount of water in it and more amount
of salt content.
We can dilute the concentrated acid but need to take certain precautions as
follows-
One can dilute concentrated acid by adding concentrated acid slowly in water with
continuous stirring. By doing so, the heat released is comparatively less and is
constantly absorbed by water. So, it prevents the reaction from becoming violent.
Bases
If we talk about bases, according to Arrhenius, they are those that when dissolved in water give hydroxide ion (OH-)
Example: Calcium hydroxide Ca(OH)2
Magnesium hydroxide Mg(OH)2
Classification of bases:
1. Classification of bases on the basis of their solubility in water
The bases that are soluble in water are called alkalis.
Note : it is interesting to note that all bases are not alkalis but all alkalis are bases.
2. Classification on the basis of strength
- strong bases
- weak bases
Strong Bases: That completely dissociates in water to give hydroxide ions. These bases totally dissociates and leave no dissociated molecule of base
Example: Hydroxides of all reactive metals
Weak Bases: That do not completely dissociates in water to give hydroxide ions.
- There actually exists the equilibrium between dissociated ions and undissociated
molecules of bases.
Example: Calcium hydroxide etc.
Class 10 Science Chapters
Physical Properties Of Acids And Bases
Some physical properties of acids are as follows –
1. They are sour.
2. They turn blue litmus red.
3. They are corrosive in nature
Some physical properties of bases are as follows –
1. They are bitter.
2. They turn red litmus blue.
3. They are soapy in nature
Chemical Properties of acids and bases
Chemical properties of acids
1. Action with Indicators: when treated with litmus, they turned blue litmus red.
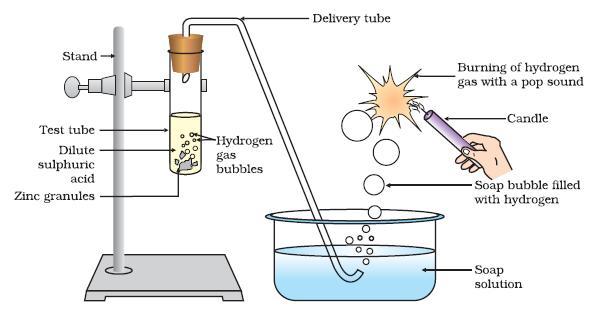
2.Reaction with Metals: They react with acid to form metal salt and hydrogen gas
is released.
Acid +metal →metal salt + hydrogen gas
Example : HCL+Zn→ZnCl2+H2
3. Action with metal carbonate and metal bicarbonate
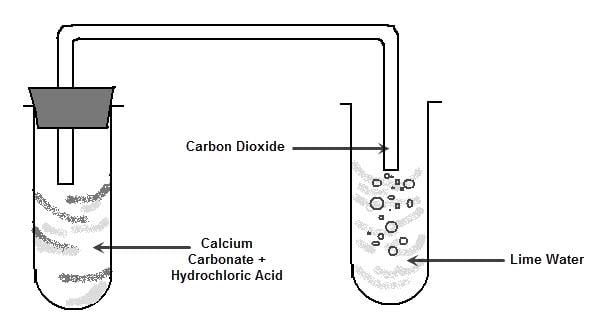
Whenever acids react with metal carbonate or metal
bicarbonate, they form respective salts, water and carbon
dioxide gas is released.
Example : acid + metal carbonate →metal salt + water + carbon dioxide gas
i.e. HCl +Na2CO3→NaCl +H2O+CO2
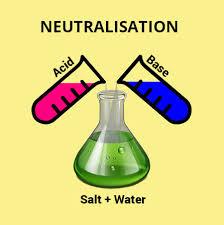
4. Reaction with Bases: Acids react with bases to form salt
and water.
i.e.
Acid + base →salt and water
HCL+NaOH→NaCl+H2O
This reaction is regarded as neutralization reaction as salt formed is neither acidic
nor basic its neutral
5. All acids act as electrolytes in aqueous states
In aqueous state when current is passed they dissociate into ions .
Chemical properties of bases
1.Action with Indicators: They turn red litmus blue
2. Reaction with Metals: They react with base to form oxy salt and hydrogen gas is released.
Example: Al+NaOH→NaALO2+H2

3. Action with ammonium salts
Example: NH4CL+NaOH→NaCL+NH4OH
4. Reaction with Acids: Acid react with acids to form salt and water
Acid + base →salt and water
HCL+NaOH—-NaCl+H2O
This reaction is regarded as neutralization reaction as salt formed is neither acidic nor basic its neutral
5. All bases act as electrolyte in aqueous states i.e. NaOH—–Na+ +OH-
6. Salts are formed when acid and base react with each other.
i.e. acid + base→salt + water
HCL+NaOH→NaCl+H2O

The salts are formed as a result of neutralization reaction as both acid and base neutralize each others effect.
The Nature of Salts Can Be-
- Acidic Salts
- Basic Salts
- Neutral Salts
Acidic Salt
It is formed when strong acid react with weak base.
As the acid is strong, therefore, strong character is retained in the salt that is why it comes out to be acidic
Example : HCL+Ca(OH)2→CaCL2+H2O
Basic Salt
It is Formed When Strong Base Reacts With weak acid
Example : NaOH+CH3COOH→CH3COONa+H2O
Neutral Salt
It is Formed When Weak Or Strong Acid And Base React With Each other. They Cancel Each Other’s Effect And Can Form Neutral Salt
Example : HCL+NaOH→NaCl+H2O
Ph and its biological importance, indicators
Strength of acid and base is expressed in terms of Ph
Ph
It is the Logarithm of Reciprocal of Hydrogen ion Concentration.
i.e.: PH =-LOG(H+)
The ph of any substance can be detected by using ph paper or ph scale.
Ph scale consist of scale ranging from 0 to 14 .
Ph ranging from 0 to 7 are acids
Ph ranging from 7 to 14 are bases
And 7 is neutral
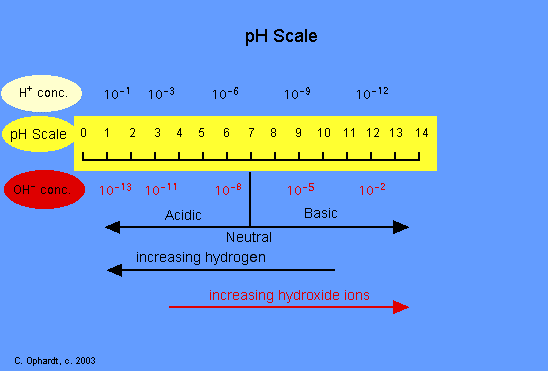
For Measuring, We Have Ph Scale Given By:
Observations Made On The Bases Of Ph Values
Ph Range Indicate the strength of acids and bases as given below :
0-2: Strong Acid
2-4: Moderately Acidic
More Than 4: Weak Acid
At 7: Neutral
7-10: Weak Base
10-12: Moderately Basic
More Than 12: Strongly Base
Effect of ph on dilution:
In Case Of Acids On Dilution :Ph Increases as acidic strength decreases
In Case Of Bases On Dilution: Ph Decreases as basic strength decreases
Importance of Ph
1. Biological Importance: Our Body Works Within 7-7.8 Ph. The Ph Of Acid Rain Is Below 5.6 And This Water Affects Aquatic Life.
2. Agriculture: Plants Also Specific Range Of Ph For Survival In Order To Grow Crops
3. In Dairies: Ph Value Of Milk Is Adjusted About 6.6. If Ph Falls Below 6.6 It Turns Sour.
4. In Tooth Decay: Tooth Decay Starts When Ph Of Mouth Falls Below 5.5. At That Ph Acid Reacts With Calcium Phosphate Of Tooth Enamel And Cause Its Corrosion. When We Eat Some Sugary Substance The Acids Production Is Stimulated That Corrode Enamel.
Indicators: Are the substances that bring about change in themselves when some acid or base is added to Natural and artificial indicators
Natural indicators: Litmus (it is extracted from lichens) original color of litmus is purple but in lab it is used as red and blue litmus.

Effect of Acid: Turns blue litmus red
Effect on Base: Turns red litmus blue
Artificial Indicators: Phenolphthalein, methyl orange
Effect on phenolphthalein-

original color is colorless
Acid: Remain colorless
Base: Turns pink
Effect on methyl orange: original color (orange)
Acid: from orange to red
Base: from orange to yellow
We have olfactory indicators as well that bring about change in smell when any substance is added to it.
Like Onion Extract And Vanilla Extract

- Effect Of Acid On Vanilla Extract: same smell
- Effect Of Base On Vanilla Extract:smell changes
- Effect Of Acid On Onion Extract:same smell
- Effect Of Base On Onion Extract: smell changes
Class 10 Science Chapters, Explanation, Notes
| Life Processes | Chemical Reaction and Equation | Carbon and it’s compound |
| Periodic Classification of Elements | Metals and Non-Metals |
Washing soda
Formula : : Na2CO3.10c
Chemical name :sodium carbonate decahydrate
Preparation : by solvays process
Reaction involved :
Na2CO3+10H2O → Na2CO3.10H2O
?The solvay?s process is an efficient process because the bi products can be reused in this process :
CaCO3→CaO+CO2
CaO+ H2O→Ca(OH)2
Ca(OH)2+NH4CL→CaCL2+NH3+H2o
This ammonia and water can be reused in solvay’s process .
Physical properties
1. It is crystalline solid
2. Action of heat :
Na2CO3.10H2O → Na2CO3+10H2O
3. Action of air : Na2CO3.10H2O → Na2CO3 H2O.+9H2O
4. Efflorescence : “the loss of 9 molecules of crystallization when washing soda is exposed to air “.
Uses of washing soda :
- Used for softening of hard water
- Used in detergents as it has cleansing properties
- Used in glass industry, etc
Baking Soda
Formula: NaHCO3
Chemical name : Sodium Bicarbonate
Preparation : by Solvay’s process
Reaction involved :
NH₃ + H₂O +NaCl + CO₂ → NaHCO₃ + NH₄Cl
Physical properties of Baking Soda:
1. It is white crystalline solid
2. action of heat : NaHCO3—HEAT→Na2CO3+H2O+CO2
uses of Baking Soda
1. it is used as antacid
2. it is used in baking cakes, etc – in cakes baking powder is added that contains baking soda and tartaric acid. in the batter of cake, baking powder is added and mixed well, then this cake is heated. On heating, the following reaction occurs –
NaHCO3—HEAT →Na2CO3+H2O+CO2
The carbon dioxide so produced, being volatile, escapes and makes the cake fluffy but the cake becomes bitter due to sodium carbonate. So, to restore bitterness, the tartaric acid reacts with sodium carbonate to form sodium tartrate that neutralizes its effect and original taste of cake is retained .
3. it is used in soda acid fire extinguishers
it consist of two cylinders –
outer: made of metal
inner: made of glass
baking soda + sulphuric acid → sodium sulphate +water + carbon dioxide
The outer cylinder has baking soda and inner one has concentrated Sulphuric acid. Both the cylinders are bounded by common knob. When knob is pressed, the glass cylinder breaks, both of them react and as a result, carbon dioxide is released that forms the protective blanket and extinguishes fire.
Plaster Of Paris
Formula :CaSO41/2H2O
Chemical name : Calcium Sulphate Hemihydrate
Preparation :
from Gypsum
CaSO₄.2H₂O —Heat 100⁰C→ CaSO₄.1/2H2O + 11/2H2O
Precaution to be taken while preparing it :
The temperature is to be controlled because if temperature exceeds 100 degree celsius, all water will get evaporated and it will form dead burnt plaster (calcium sulphate)
CaSO4.2H2O—Temp. above 100→CaSO4+2H2O
Physical Properties of POP
- white crystalline solid
- Reaction with water : when used it has to mixed with water and following reaction occurs-
POP + H2O → Gypsum
Uses of POP
- it is used to decorate ceilings, etc
- It is used to make statues, etc
- It is used for joining fractured bones
Bleaching Powder
Formula : CaoCL2
Chemical name :Calcium Oxychloride
Preparation :
Ca(OH)₂ + Cl₂ → CaOCl₂ + H₂O
Physical properties of Bleaching Powder
1. white crystalline solid
2. Action of air : CaoCL2+CO2—-CaCO3+CL2
Uses of Bleaching Powder:
- it makes wool unshrinkable
- It is used for cleaning water tanks ,toilets, etc.
Important Videos Links
Acids Bases and Salts NCERT Solutions
Q1. A solution turns red litmus blue, its pH is likely to be
(a) 1
(b) 4
(c) 5
(d) 10
A : 10
Q2. A solution reacts with crushed egg-shells to give a gas that turns lime-water milky. The solution contains
(a) NaCl
(b) HCl
(c) LiCl
(d) KCl
A :NaCl
Q3. 10 mL of a solution of NaOH is found to be completely neutralised by 8 mL of a given solution of HCl. If we take 20 mL of the same solution of NaOH, the amount HCl solution (the same solution as before) required to neutralise it will be
(a) 4 mL
(b) 8 mL
(c) 12 mL
(d) 16 mL
A : 16ml
Q4. Which one of the following types of medicines is used for treating indigestion?
(a) Antibiotic
(b) Analgesic
(c) Antacid
(d) Antiseptic
A : antacaid
Q5. Write word equations and then balanced equations for the reaction taking place when – (a) dilute sulphuric acid reacts with zinc granules.
(b) dilute hydrochloric acid reacts with magnesium ribbon.
(c) dilute sulphuric acid reacts with aluminium powder.
(d) dilute hydrochloric acid reacts with iron filings.
A :Zn +H2SO4–>ZnSO4+H2
Mg +2HCL–>MgCL2+H2
2Al +3H2SO4–>Al2(SO4)3+ H2
2Fe+6HCl–>2FeCL3+3H2
Q6. Compounds such as alcohols and glucose also contain hydrogen but are not categorised as acids. Describe an Activity to prove it.
A :Alcohols and glucose when added to litmus show no effect because they do not get ionized when added to water. Therefore, have no effect on litmus.
?
Q7. Why does distilled water not conduct electricity, whereas rain water does?
A :Distilled water not conduct electricity, whereas rain water does because distilled water is pure form of water so it does not ionize whereas rain water contains impurities that makes it good conductor of electricity.
?
Q8. Why do acids not show acidic behaviour in the absence of water?
A: Acids not show acidic behaviour in the absence of water because in absence of water they do not get ionized to produce hydrogen ions
Q9. Five solutions A,B,C,D and E when tested with universal indicator showed pH as 4,1,11,7 and 9, respectively. Which solution is
(a) neutral?
(b) strongly alkaline?
(c) strongly acidic?
(d) weakly acidic?
(e) weakly alkaline?
(f) Arrange the pH in increasing order of hydrogen-ion concentration.
A: (a) 7
(b) 11
(c) 1
(d) 4
(e) 9
(f) 11<9<7<4<1
Q10. Equal lengths of magnesium ribbons are taken in test tubes A and B. Hydrochloric acid (HCl) is added to test tube A, while acetic acid (CH3COOH) is added to test tube B. Amount and concentration taken for both the acids are same. In which test tube will the fizzing occur more vigorously and why?
A. Fizzing will occur more vigorously in test tube B because in it carbon dioxide is evolved that escapes with fizz .
Q11. Fresh milk has a pH of 6. How do you think the pH will change as it turns into curd? Explain your answer.
A: Fresh milk’s Ph decreases as the bacteria in milk starts converting lactose into lactic acid
Q12. A milkman adds a very small amount of baking soda to fresh milk.
(a) Why does he shift the pH of the fresh milk from 6 to slightly alkaline?
(b) Why does this milk take a long time to set as curd?
A : (a)To prevent rancidity
(b) Because it takes more time for lactobacillus to grow and change milk to curd
Q13. Plaster of Paris should be stored in a moisture-proof container. Explain why?
A :Plaster of Paris should be stored in a moisture-proof container because if it comes in contact with air, it is transformed into hard substance called gypsum.
Q14. What is a neutralisation reaction? Give two examples.
A : Neutralization reaction : a reaction in which two compounds react to form neutral substance .
i.e., ACID +BASE →SALT +WATER
HCl+NaOH→NaCl+H2O
Q15. Give two important uses of washing soda and baking soda.
A : washing soda :
- It is used for softening of hard water
- It is used in glass industry
Baking soda :
- It is used for making idlis ,cake, etc
- It is used in making fire extinguisher
Extra Questions And Answers
1.State Arhenius theory?
Ans. According to him, acids are the substances which when dissolved in water give H3O+ ions. Bases are those substances which when dissolved in water gives OH- ions.
2. What is the difference b/w dilute HCl & concentrate HCl?
Ans.In dilute HCl → amount of H2O is more
In conc. HCl → amount of H2O is less.
3. What is the effect of acid on litmus, Phenolphthalein?
Ans.Acid turns litmus from blue to red & phenolphthalein from pink to colourless.
4.Write Ph of pure water?
Ans.Ph of pure water is ‘7’
5.What is the minimum & maximum value of PH meter?
Ans.Minimum value of Ph meter is 0 and Maximum value is 14.
6.Differentiate between base and alkali.
Ans.
|
base |
alkali |
|
a substance which on dissolving in water gives OH ion |
a soluble base is called an alkali |
|
all bases are not alkalis |
all alkalis are bases |
|
Example – Mg(OH)₂ |
Example – NaOH |
7. Write reaction of an acid with metal oxide, metal hydroxide?
Ans. NaOH + HCl → NaCl + H2O (neutralization )
8. What do you mean by basicity?
Ans. Basicity is defined as “Number of ions produced when acid is dissolved in water”. For example – H2SO4 → (dibasic)
9. To which category acetic acid belongs?
Ans. Acetic acid belongs to Organic acid & is week acid.
?
10.Write the effect of dilute HCl on Na2CO3?
Ans. Na2CO3 + HCl → NaCl + H2O + CO2
11. What is neutralization reaction?
Ans. A reaction of an acid and base to form salt & water is called a neutralization reaction.
Example – HCl + NaOH → NaCl + H2O
12.Why sulphuric acid is called acid?
Ans.Sulphuric acid is called acid because when dissolved in water, it dissociates to give H3O+ ions.
13.Which of the following hydroxide is not alkali? NH4OH, Ca+(OH)2 + Cu-(OH)2 NaOH?
Ans.Cu (OH)2 & Ca(OH)2 are not alkali.
14. Name is organic acids?
Ans. HNO3, H2SO4 & HCl are organic acids.
15. Name king of chemicals?
Ans. H2SO₄ is called the king of chemicals.
16.Out of phosphoric & carbonic acid which is weak acid?
Ans. Both phosphoric & carbonic acid are weak acids.
17.What is the value of ‘x’ for sling acids?
Ans. The value of ‘x’ for sling acids is more than 30 % .
18.Name the acid present in lemon, tomato & curd?
Ans. Lemon contains citric acid.
Tomato contains oxalic acid.
Curd contains Lactic acid.
19.What is the nature of metal oxide & Non metal oxides?
Ans. Metal oxides are Basic while non-metal oxides are acidic.
?
20.Out of KOH, Ba(OH)2 , Ca(OH)2, NaOH, pick weak oxide?
Ans. Ca (OH)2
21. What is the original colour of litmus & phenolphthalein?
Ans. Litmus (purple) , phenolphthalein (colourless)
22. Name the plant & its division for which litmus can be extracted?
Ans. Lithens & div- thelcphyte
23. What is the effect of dry HCl gas on dry litmus paper?
Ans. No effect : dry HCl does not produce H+ ions
24. Write any two application of neutralization reaction?
Ans.A) it can remove acidity of oil
B) it helps astronauts in reducing hamful levels of Co2.
25. Name the substance that is added to sail to remove its acidity?
Ans.Slaked lime i.e. (Ca(OH)2) is added to salt to remove its acidity.
?
26.How sting of yellow weeps can be neutralized?
Ans. Sting of yellow weeps can be neutralized by rubbing acetic acid (vinegar).
?
27.When acid/base is mixed with water, concentration of H+/oH- decreases or increases?
Ans. Concentration of H+/oH- decreases on dilution.
28.Write the effect of bases with methyl orange?
Ans.Bases turn methyl orange to yellow.
29. Complete – NaOH + Al → NaAlO2 + ___
Ans. NaOH + Al → NaAlO2 + H2
30. Complete NH4Cl + NaOH → NaCl + _____ + H2O
Ans. NH4Cl + NaOH → NaCl + NH3 + H2O
31.Why bases act as electrolyte?
Ans. Bases act as electrolyte because they produce ions in solutions.
?
32.Write one use of Ca(OH)2 & NaOH?
Ans. Ca(OH)2 is used in preparing bleaching powder
NaOH is used in preparing soaps
33.Which gas is liberated when acids react with metal carbonates?
Ans. CO2 is liberated. It turns lime water milky
34.Define Ph.
Ans. Ph is the Logarithm of Reciprocal of Hydrogen ion Concentration.
35. Write the value of Ph at which our body works?
Ans.Our body works at a Ph level of 7.0 & 7.8 .
36. When rain water forms acid rain?
Ans . When the Ph of rain water goes lower than 5.6, it becomes acid rain.
?
37. How do we come to know that milk has turned sour?
Ans. When the Ph of milk becomes lesser than its normal Ph i.e. 6.6, it turns sour.
38. At what Ph tooth decay starts and why?
Ans. Tooth decay starts at a Ph of below 5.5 ; excess of acid reacts with enamel (Ca3Po4) & corrodes it.
39. How one can be relieved from the sting of beetle?
Ans. One can be relieved from the sting of beetle by rubbing it with the leef of dock plant. We feel the sting due to the secretion of methanoic acid from it.
Important Questions Videos Links